DISCUSSION
Until now, only 3 species of mites and a flea species were reported from red squirrels in Korea [
3-
5]. In this study, we found 3 species of ectoparasites and a species of tapeworm. With the exception of
C. (
Monopsyllus)
indages, 3 other species (
C. dendritica,
H. sciurinus, and
L. pallidum) were firstly detected from the red squirrel in Korea.
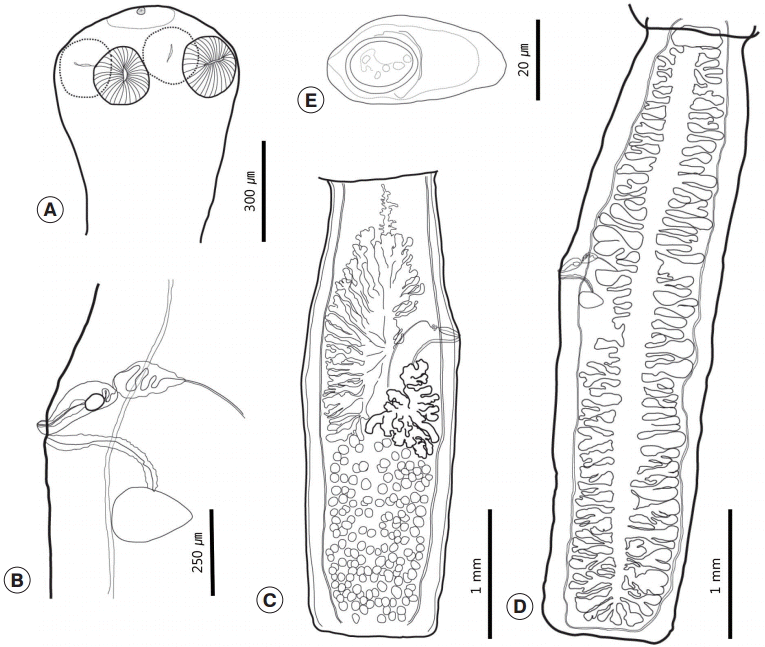
Catenotaenia tapeworms are parasites of various rodents that belong to 3 suborders (Myomorpha, Sciuromorpha, and Castorimorpha) [
6]. Haukisalmi et al. [
6] provided a taxonomical review of the genus
Catenotaenia in Eurasia; they sought to distinguish each species using morphological characteristics, including the outline shape of the segments and 28s rRNA sequencing. According to their review, our specimens appear morphologically similar to
C. dendritica.
C. dendritica is known to be a host-specific parasite of
S. vulgaris, and, indeed, our specimens were recovered from this host. This species has several distinct characteristics compared with other
Catenotaenia species. First, the proglottids are elongated and have an almost uniform width. Second, there are numerous uterine branches. Third, the testicular field is divided longitudinally [
6]. In addition, as reported by Joeux and Baer [
7],
C. dendritica has elongated proglottids, and the length/width ratio; the mature and gravid proglottids are 4 and 8 times longer than the width of the proglottid, respectively. Spaskii [
8] also noted that the proglottid width varied very little; he termed it as ‘almost constant’. Moreover, he considered the longitudinal division of the testicular field and the number of testes (<300) to be key features of this species [
8]. Haukisalmi et al. [
6] also suggested additional diagnostic characteristics, namely, the presence of the anterior free space, comparatively short ovary (<40%), antero-poral merging of the seminal receptacle and vagina, and position of the seminal receptacle (positioned anterior to the Mehlis’ gland). All of these morphological features were also observed in our specimens.
However, our specimens also matched to the description of
Catenotaenia ris Yamaguti, 1942. Yamaguti [
9] obtained several specimens from the small intestine of
Sciurus lis Temminck from Nagano Prefecture, Japan and named the species
Catenotaenia ris. He found that his species differed from
C. dendritica in that its apical sucker was small relative to the 4 lateral suckers, and there were fewer testes [
9]. In relation to the number of testes, Yamaguti [
9] reported 140-190 testes in
C. ris, whereas Riggenbach [
10] reported that
C. dendritica had 200-210 testes. However, when Joyeux and Baer [
7] re-observed the Riggenbach’s material, they found that the real number of testes was 140-190. Our specimens had 133-178 testes. In addition, Joyeux and Baer [
7] described the degeneration of the apical sucker in adult
Catenotaenia pusilla (Goeze, 1782), although it appeared to be functional in metacestodes. On the basis of these observations, they considered
C. ris as a synonym of
C. dendritica [
7]. This conclusion was supported by Spasskii [
6] and Haukisalmi et al. [
8]. This suggests that our specimens are indeed
C. dendritica. However, it should be noted that the morphometric data of
C. dendritica provided by Haukisalmi et al. [
6] exhibit considerable variation, especially in terms of the number of testes (130-233) and uterine branches (35-60). This suggests that either their data were from at least 2 different species or their data included erroneous data, similar to the case of Riggenbach [
7]. In any case, we considered our specimens to be
C. dendritica at this time, but this conclusion is tentative and will have to be confirmed by additional molecular analysis and comparison with the known sequences of
C. dendritica [
6].
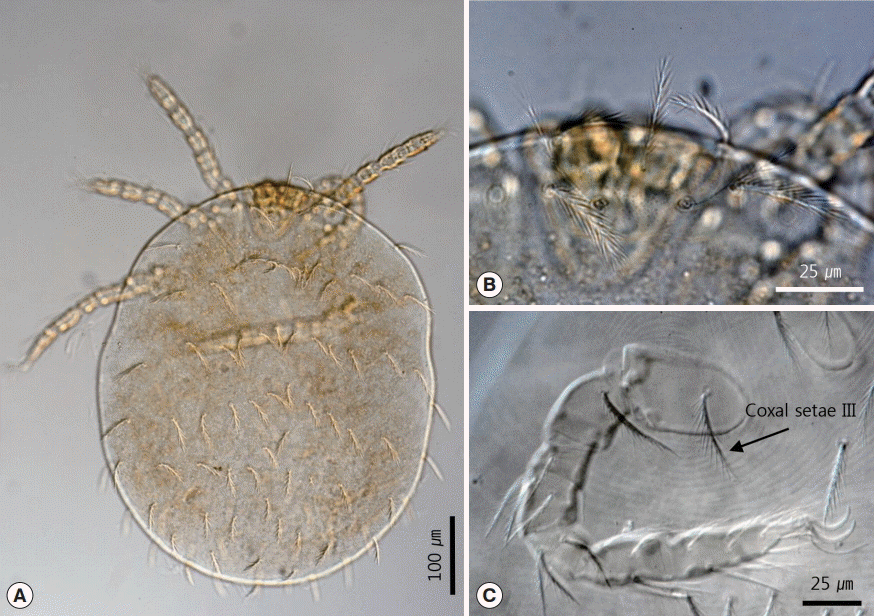
Unfortunately, we could not collect male mesostigmatid mites. Thus, our morphological observations were based on female specimens only. However, this seems sufficient for identifying the species. Three species of
Hirstionyssus mites are known to distribute in Korea, namely,
H. isabellinus (Oudemans, 1913),
H. sunci Wang, 1962 (=
H. apodemi Zuevsky, 1970), and
H. carnifex (Koch, 1829) [
4,
11,
12]. Kim et al. [
11] also found males, females, and deutonymphs of mesostigmatid mites in
Rattus norvegicus and
Apodemus agrarius, and identified these as
H. apodemi. However, Uchikawa [
13] concluded that
H. apodemi is a synonym of
H. sunci after observing the morphological variation of the sternal shield. This conclusion was supported by Mašán and Fenda [
14]. We also agree with this conclusion and thus call this species
H. sunci in the present report. However, our specimens did not match the descriptions of the 3
Hirstionyssus mite species reported previously in Korea [
11,
12]. Unlike these
Hirstionyssus mites, our specimens had a tapered subhexagonal dorsal shield and a sternal shield that was longer than st1. Although
Hirstionyssus carnifex also has a subhexagonal dorsal shield, the posterior region of its shield is wider than the anterior region [
14]. Its sternal shield also looks similar to that in our specimens, but it is wider.
Hirstionyssus sunci and
Hirstionyssus isabellinus differ from our specimens by having a suboval-shaped dorsal shield and a longitudinally narrow sternal shield because of the curved posterior margin. In particular, our specimens differed from these 3
Hirstionyssus mite species most markedly in their coxal spur formula; it was 0-3-2-0 in our specimens, whereas it is 0-2
(1+1)-2
(0+2)-0 in
H. isabellinus, 0-2
(1+1)-2
(0+2)-1
(0+1) in
H. carnifex, and 0-2
(1+1)-2
(0+2)-1
(0+1) in
H. sunci [
14]. Thus, on the basis of these morphological differences, we conclude that our specimens are a previously unrecorded mite species in Korea. Instead, it is likely that our specimens belong to
H. sciurinus since the morphological characteristics of our specimens perfectly match those of
H. sciurinus from Slovakia [
14] and the original description [
15]. Since this species is also known to be a parasite of the red squirrel in Slovakia [
14,
16], we regard our specimens as being
H. sciurinus.
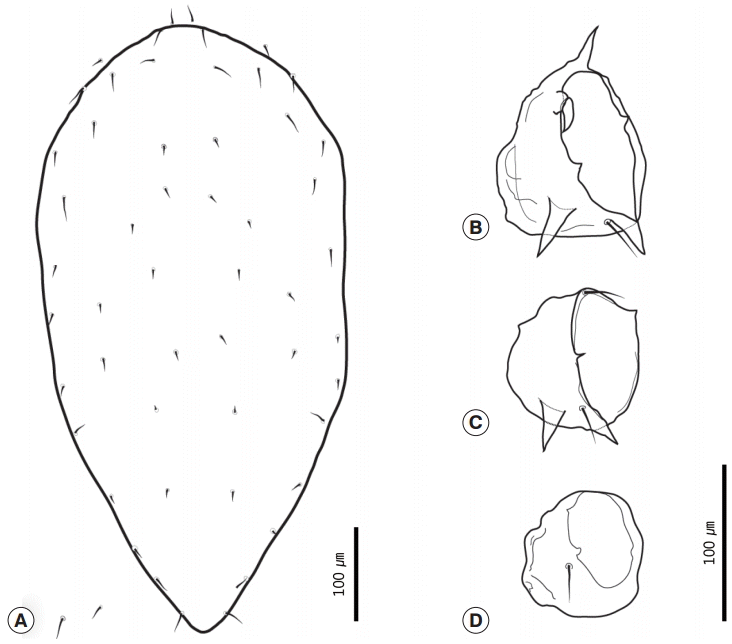
Comparison with the previously suggested key for the chigger mites [
17] indicated that these characteristics were sufficient to identify our specimens as
L. pallidum. This species is known to be a predominant species in small mammals from the northern part of Korea [
18-
20]. Several records indicated that larval
L. pallidum has a variety of hosts [
3,
18-
21]. However, the red squirrel has not been reported previously to be a host for this ectoparasite, although there is a report that chigger mites (including
L. pallidum) have been found in
S. v. lis in Japan [
22]. Thus, the present study suggests that the red squirrel may be another host of chigger mites in Korea.
In the present study, fleas were the most dominant ectoparasites of the red squirrels. Only 1 species,
C. (
Monopsyllus)
indages, has been reported in red squirrels in Korea [
4]. The specimens in our study also had all the morphological characteristics of
C. (
Monopsyllus)
indages that were described previously [
23,
24]. The species was first reported by Rothschild [
23] on the basis of the morphology of female fleas collected from
S. vulgaris orientalis in Hokkaido, Japan.
Of the parasite species investigated in the present study,
C. dendritica seems to be the least important in terms of human health whereas arthropod parasites may cause health problems. Many fleas are known to lack strict host selection, and this means that fleas can readily act as vectors of zoonotic diseases [
25]. In addition, several human cases of rat mite dermatitis caused by the mesostigmatid mite
Ornithonyssus bacoti have been reported in Korea [
26]. It is unclear whether
H. sciurinus can bite humans. However, it is quite possible that it, like other mite species, can act as an allergen. In the case of chigger mites,
L. pallidum is known to be a vector of tsutsugamushi disease in Korea, which is caused by
Orientia tsutsugamushi: this was proven by Rhee et al. [
27] who found by an indirect fluorescent antibody test that 2 of 447 individual
L. pallidum collected in Korea bore
Orientia tsutsugamushi. Although we did not try to identify the pathogens in either our specimens or the hosts, the fact that our red squirrels carried
L. pallidum suggests that the red squirrel in Korea could potentially act as a reservoir host for tsutsugamushi disease. Although ectoparasite infection in Korea is generally regarded as an occupational infection of farmers or people who associate with fields, our results suggest that ectoparasites may be unexpectedly close to the sidewalks and lawns that are frequently used by people. Thus, caution may be needed when people undertake outdoor activities in places that are inhabited by animals like the red squirrel, even university campuses.
Compared with the previously reported parasitic fauna of red squirrels, our squirrels had relatively poor parasitic species diversity. Feliu [
28] found 5 species of helminths in 248
S. vulgaris from the Iberian Peninsula, namely,
C. dendritica,
Trichostrongylus retortaeformis, Mastophorus muris, Syphabulea mascomai, and
Rodentoxyuris sciuri. Moreover, Shimalov and Shimalov [
29] found 11 species of helminths in 30 adult specimens of
S. vulgaris in Belorussian Polesie. Cyprich et al. [
16] recorded 19 ectoparasite species in 93
S. vulgaris specimens and 32 nests in Slovakia. Although some of the species were considered occasional infections, the species variety was larger than that in our squirrels. Romeo et al. [
30] found 5 species of nematodes and 13 specimens of unidentified cestodes in 147 red squirrels; they also found 7 species of ectoparasites in 311 squirrels [
30]. We only found 1 tapeworm and 3 species of ectoparasites. Moreover, we could not find any of the previously reported mite species of
S. vulgaris in Korea [
3,
5]. There are several possible reasons for this. First, Romeo et al. [
30] suggested that the macro-parasitic fauna of red squirrels may be mainly composed of parasites that have direct life cycles because the red squirrel is an independent animal that spends almost all of its time in trees [
31]. The poor species diversity that we observed may also reflect habitat fragmentation. The study area was surrounded by broad roads that isolated the area from other red squirrel habitats such as mountains and woodlands. In addition, the red squirrel population in the area had been estimated to include fewer than 30 individuals (personal communications). This environmental isolation may interrupt the exchange of parasites between individuals and also other populations, and even other species. In addition, carnivores and ungulates are absent in the area or clearly rare. The absence of these kinds of animals could decrease the chance that the squirrels become infested with ticks or species of helminths that use these animals as the final host. Moreover, present study has a limitation of collecting ectoparasites because they tend to escape from its host when the host is dead. Therefore, to improve our understanding of the parasitic fauna of the red squirrels and their ecology in Korea, further studies are needed with animals collected from well-preserved natural environments.
In conclusion, the present study investigated the parasitic fauna of red squirrels in a university campus in Korea. We added 2 previously unrecorded species, C. dendritica and H. sciurinus to the Korean taxa and showed that the red squirrels may carry ectoparasites of public health importance.