INTRODUCTION
Oculotrema hippopotami Stunkard, 1924 (Oculotrematinae Yamaguti, 1968), the only monogenean known for successful colonization on a mammal poses many unanswered questions. It was originally described from
Hippopotamus amphibius Linnaeus, 1758 from the Giza Zoo (Egypt), but has also been reported from Sudan and Uganda [
1], Zimbabwe [
2] and South Africa [
3,
4]. Stunkard gave a comprehensive description of
O. hippopotami, though based on highly contracted specimens [
5]. Du Preez et al. [
4] amended the original description of an adult by adding information on size of the parasite, particulars of egg and copulatory system structure, redefined morphological parameters of adults, and revealed the presence of a bucco-oesophageal canal, a uterine evagination, an operculated egg, the retention of marginal hooklets in the mature parasite and the ability to double its length and feed over a large area around its position of attachment. They reported a maximum of 24 parasites on a single eye and a maximum of 37 on an individual host specimen. The prevalence was found to be 75% for adults, 85% for sub-adults and 90% for the total sample [
4]. In addition, Du Preez et al. and Tinsley [
4,
6] studied oncomiracidia and Thurston studied the larvae of
O. hippopotami [
7]. Sub-adults of
O. hippopotami were briefly mentioned in a few publications but never were studied exclusively. Although Thurston [
7,
8] and Du Preez et al. [
4] mentioned immature worms (sub-adults), but none provided more details about them.
Some studies have been devoted to the habitat of this monogenean, the hippopotamus eye tissues, but their histopathological changes at the place of attachment of the parasite have never been studied. The same applies to details of the distal parts of the copulatory system that have not been studied using scanning electron microscopy (SEM) [
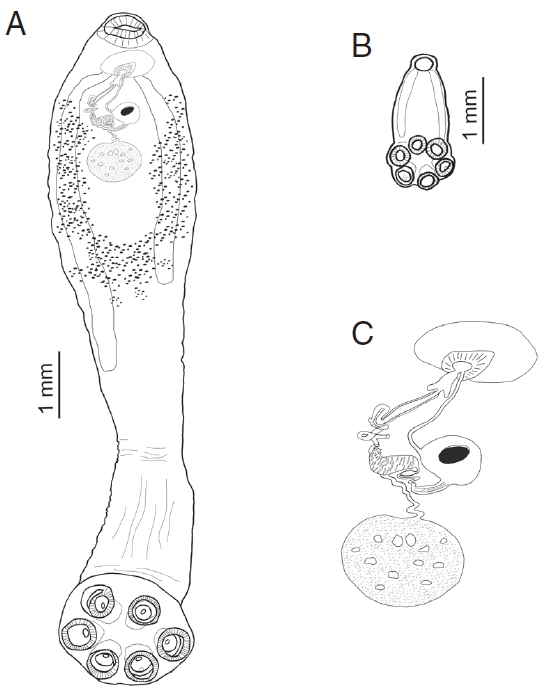
9]. To date there are no comparative studies on morphological and metrical characteristics of adults and sub-adults of
O. hippopotami. In addition, this is the first study on the structural analysis of this parasite comparing adult and sub-adult forms and providing further information on this unique mammalian monogenean.
DISCUSSION
Because of severe drought in August and September 2016 in northeastern South Africa and declining overall condition of hippopotami, a decision was made to control compromised animals from the Kruger National Park Associated Private Nature Reserves (APNR).
The present infection levels of
O. hippopotami compare to that reported in the previous studies, and are very high and unique in Monogenea under natural conditions compared to almost all other representatives of Monogenea in general [
15], and particularly Polystomatidae [
16]. Five of 6 examined animals from the current study were infected. Thurston & Laws reported a prevalence of 76% on a large sample size (960 of 1,263 hippopotami were positive for
O. hippopotami) [
1]. Du Preez et al. [
4] reported a prevalence of 90%. Several factors probably contribute to this phenomenon. Life in clusters ensures fertilization, and, consequently, the release of eggs into the water. A pronounced care for the offspring, the constant contact of the mother with the calf, and the presence of adult individuals in the proximity to the standing water ensures infection. The only serious obstacle is the mechanical influence on the worms of the eyelid during blinking, which they successfully overcome by developing a unique sclerotized suction attachment system. The degree of attachment to the tissues of the host is so strong that in a number of cases, it was practically impossible to tear off the monogeneans from the eye tissue. High levels of infection together with absence of obvious pathogenicity to the host indicate an extremely successful adaptation of this parasite to the host. The effect of the infection of this monogenean on the host does not appear to have an influence on visual acuity, since no studies indicated the presence of the
O. hippopotami specimens in the anterior part of the eye or cornea.
New morphological features were revealed in the present study, such as flattened dorsal side of the body provided for the first time with SEM micrographs; the presence of genital papillae observed only in adults. In addition, differences were shown in the orientation of oral suckers between adults and sub-adults. The new information on sperm ejaculatory structures of O. hippopotami was shown with SEM micrographs. New metrical and graphical information obtained for adults and sub-adults were compared with previous studies. Finally, the metrical data on the distal part of the vas deferens is reported for the first time.
Halton and Jennings stated that specimens of
O. hippopotami feed on epithelial cells and mucous [
17].
Fig. 3B shows an adaptation of the whole body shape to this mode of nutrition - flattened ventral side of the body contrasting with arched dorsal side. Earlier, Moeng et al. [
18] reported that the oral sucker is designed for long-term positioning of the worm, clamped between the host’s eyeball and eyelid. It was reported that the oral sucker morphology reflects the mode of feeding. The sub-terminal flat mouth of
O. hippopotami was considered to scoop up the mucous and epithelial cells [
18]. The musculature of oral sucker of adults is more developed than in sub-adults; compare
Fig. 3C and
Fig. 4B. The size of the worm suggests that increased food consumption causes greater development of the musculature of the oral sucker. Tegument structure revealed in the present study has a typical polystomatid honeycomb-structure (
Fig. 4E) [
19].
According to Tinsley [
6], Du Preez et al. [
4] and our observations, specimens of
O. hippopotami do not possess a vagina, however Theunissen, mentioned its presence and marked it on a photograph (
Figs. 2,
4,
7 pp 125) [
9]. The cirrus of
O. hippopotami has no armament [
6] and because of this unarmed cirrus, hypodermal insemination that has been reported in other monogenean groups does not appear to be the case for
O. hippopotami [
4] and the process of insemination for this species remains unknown. Du Preez et al. [
4] proposed that in
O. hippopotami the sperm is introduced via the muscular cirrus directly in the genital atrium. Using SEM in the present study (
Fig. 3C, D) we could show the muscular cirrus and ejaculated sperms; ejaculation possibly was triggered due the thermal or chemical effects during fixation of living worms in ethanol. Similar to the event observed in other groups of monogeneans (Diplozoidae), where placing living or recently dead monogeneans in a drop of glycerin-gelatin on a slide, triggers the ejection of the contents of the digestive tract as observed with a stereomicroscope (Rubtsova, unpublished data). The genital pore is surrounded by prominent muscular papilla (
Fig. 3C, E), noted in all adult specimens, that may possibly play an important role in the process of insemination.
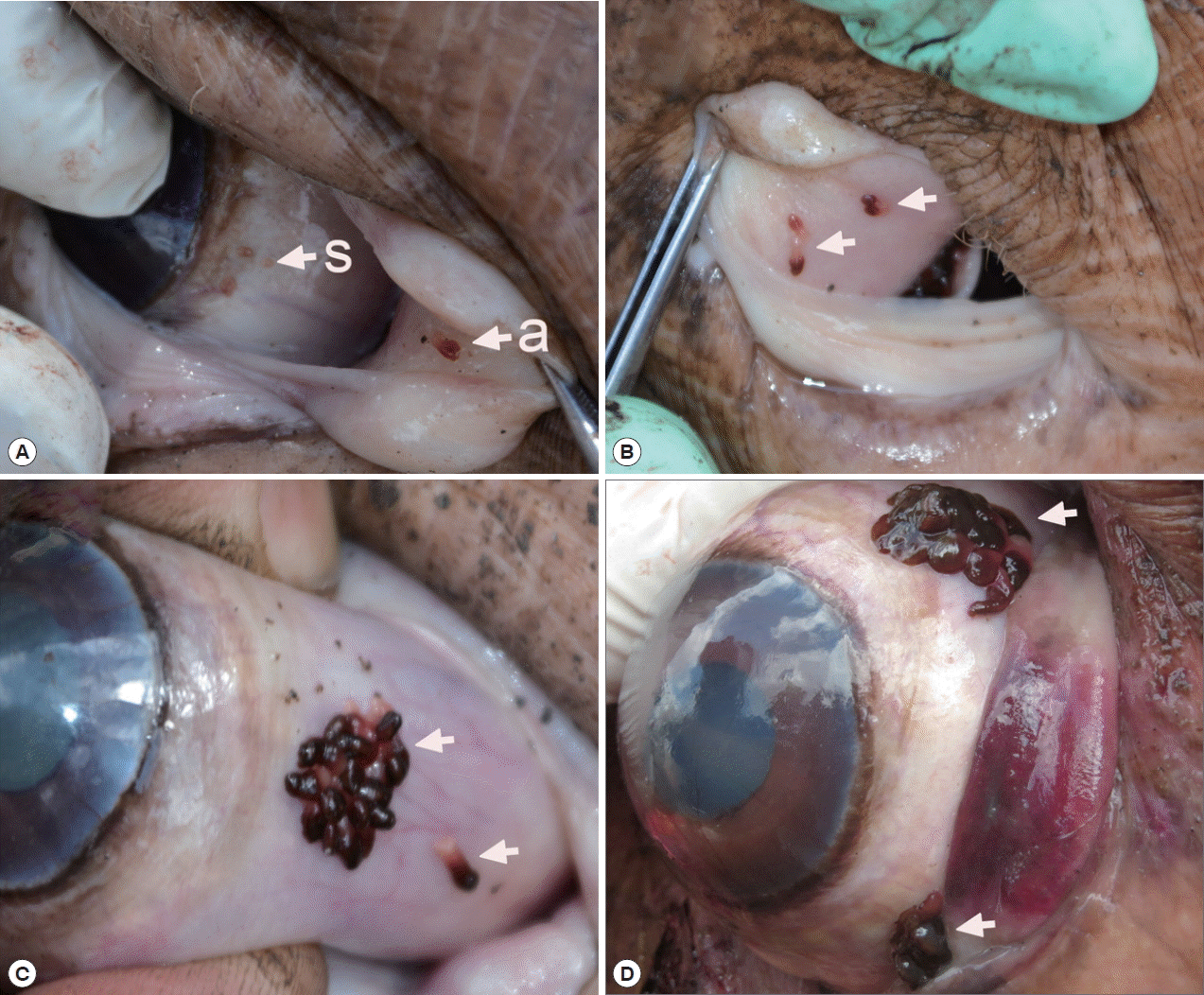
All infected hosts of the present study were infected with either adult worms, or a combination of adults and sub-adults. Our observations (see
Fig. 1A), as well as observations of Du Preez et al. [
4] (see
Fig. 1B of the latter authors), demonstrate that the same individual hippopotamus can be infected by different generations of monogeneans during its lifespan. Moeng observed the same tendency [
20]. Therefore, the assumption can be made that the presence of a mixture of different generations of
O. hippopotami is a common occurrence in the eyes of hippopotamus as was noted during the current study in 3 of 6 studied hosts. This feature is more advantageous for the survival of the parasites. For representatives of
Polystoma genus, on the contrast, it is typical to see only one age group present in the urinary bladder of one anuran amphibian host [
19,
21].
It was previously noted by Du Preez et al. [
4], as well as seen during the present study, that
O. hippopotami spend their lifespan in clusters or groups in certain part of the hippo’s eye. Sub-adults found on the same eye together with adults somehow settle away from the adult clusters. This may be related to the necessity of mutual fertilization of individuals of one cluster. Adults appear to stay together in clusters during their lifetime. Sub-adults usually found separately from the cluster of adults and may begin a new colony and not compete for the resources with parental specimens (
Fig. 1A(s)), also see
Fig. 1B in Du Preez et al. [
4].
The size differences were the most reliable characteristics for differentiation of adult and sub-adult forms of polystomatid monogeneans [
21]. Measurements of body size of sub-adults in the present study (
Table 2) are comparable to those of adults described by Stunkard [
5]. The presence of eggs and the level of development of the reproductive system (dimensions of uterus, ovary and testes) of Stunkard’s [
5] specimens correspond to the dimensions mentioned by Du Preez et al. [
4] and our data for adults. Differences in body size may be due to extremely contraction of the worms as mentioned in the original description [
5].
In our study, the average body length of an adult was 3.5 times larger than that of sub-adults. Note that the length of the body for
O. hippopotami is very variable due to its ability to contract. As was noted in the present study, the tegument, that has a honeycomb-structure, consists of plates elastically connected to each other (
Fig. 4D) with strands between plates. This structure allows the body of
O. hippopotami to stretch freely in length and shrink almost into a sphere as observed during this study. Considering the maximum measurement of body length from Du Preez et al. [
4] for an adult being 32.51 mm and minimal length of sub-adults in our study is 1.2 mm, this ratio could reach 27.
The relative size of the suckers to the size of the haptor varies (3.2 in adults to 2.9 in sub-adults), but not as significantly as the ratio of the size of the haptor to the length of the body (6.8 in adults to 3.7 in sub-adults).
Most of the measurements of adults are within the variability range specified in previous publications, except for a notable difference in body length and width in the original description that is noticeably smaller than those described in Du Preez et al. [
4] and the present study. We also report herein the length of a thick-walled muscular portion of the vas deferens and the diameter of the cirrus for both adults and sub-adults for the first time.
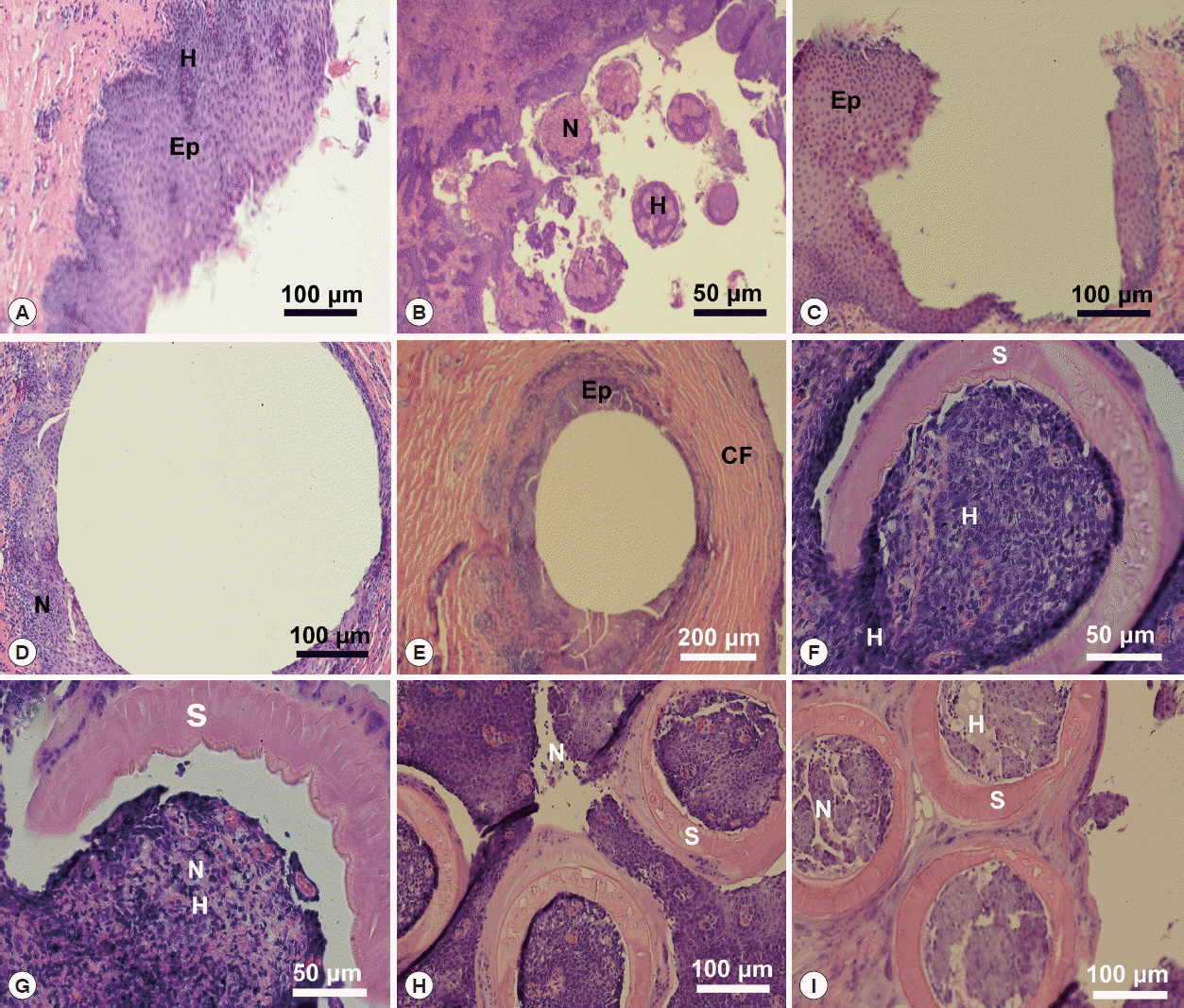
Du Preez et al. [
4] mentioned the prominent bud-shaped attachment marks left after the removal of the parasite. The present study indicates a convexity of host tissue is grabbed inside the sucker. Pathological changes in the host tissues included necrosis and hemorrhage at the sites of attachment of the parasite. See
Fig. 5 for pathological changes in the host tissue at attachment sites that are due to the mode of the attachment of
O. hippopotami. The suction is aided by skeletal musculature and reinforced with an internal skeleton of the suckers [
6]. During its evolution,
O. hippopotami lost its hamuli, due to the delicate habitat, where irritation of host tissue could be disadvantageous [
6]. It is quite possible that such kind of influence does not have a pronounced negative effect on the functions of the host as a whole, but mostly by putting a burden on the tissue immunity. Most likely the presence of
O. hippopotami in the eye (both on the side of the globe or on nictitating membrane (but never on cornea) does not affect the visual acuity of the hippopotamus. Similar changes in attachment points are also known for other groups of parasites, for example, acanthocephalans [
23].
Such pathological changes, i.e. necrosis and hemorrhage found at the host tissues, cause a slight deterioration in the quality of life of the host (parasite, obviously never is an obstacle to the eyesight acuity of the host). Thus, this parasite-host system is an example of an almost ideal mutual adaptation, and O. hippopotami can be considered one of the most successful monogeneans.
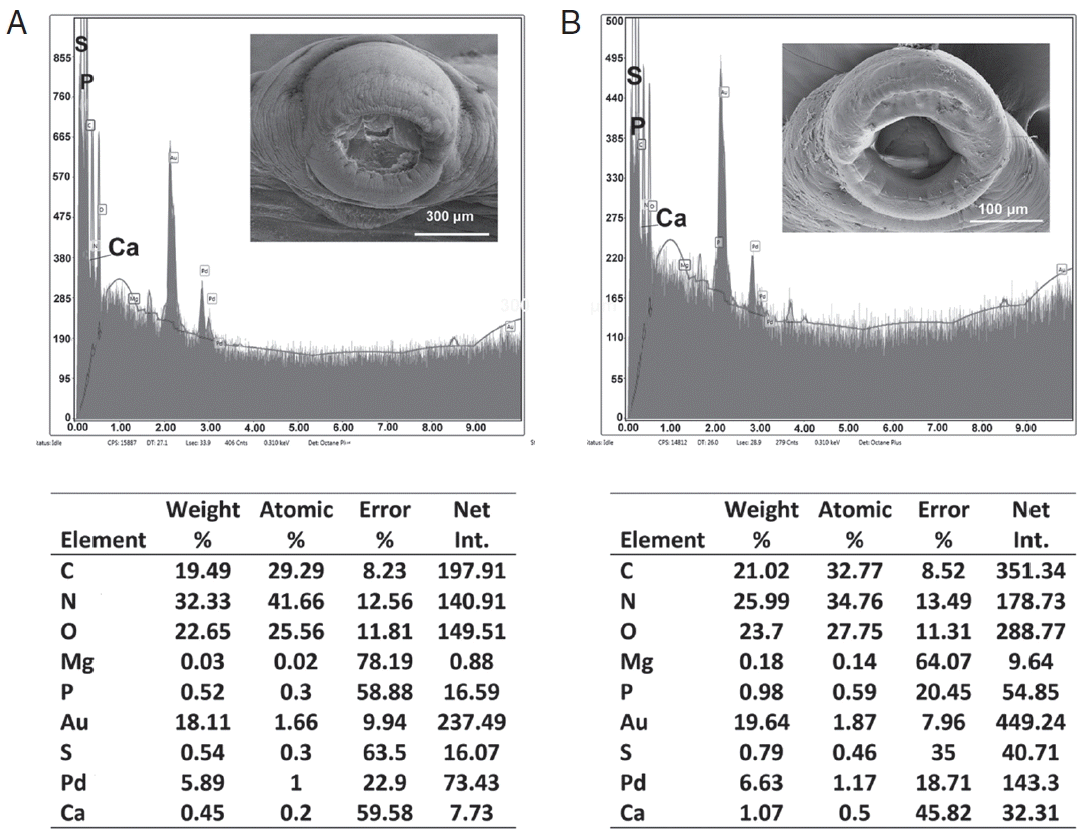
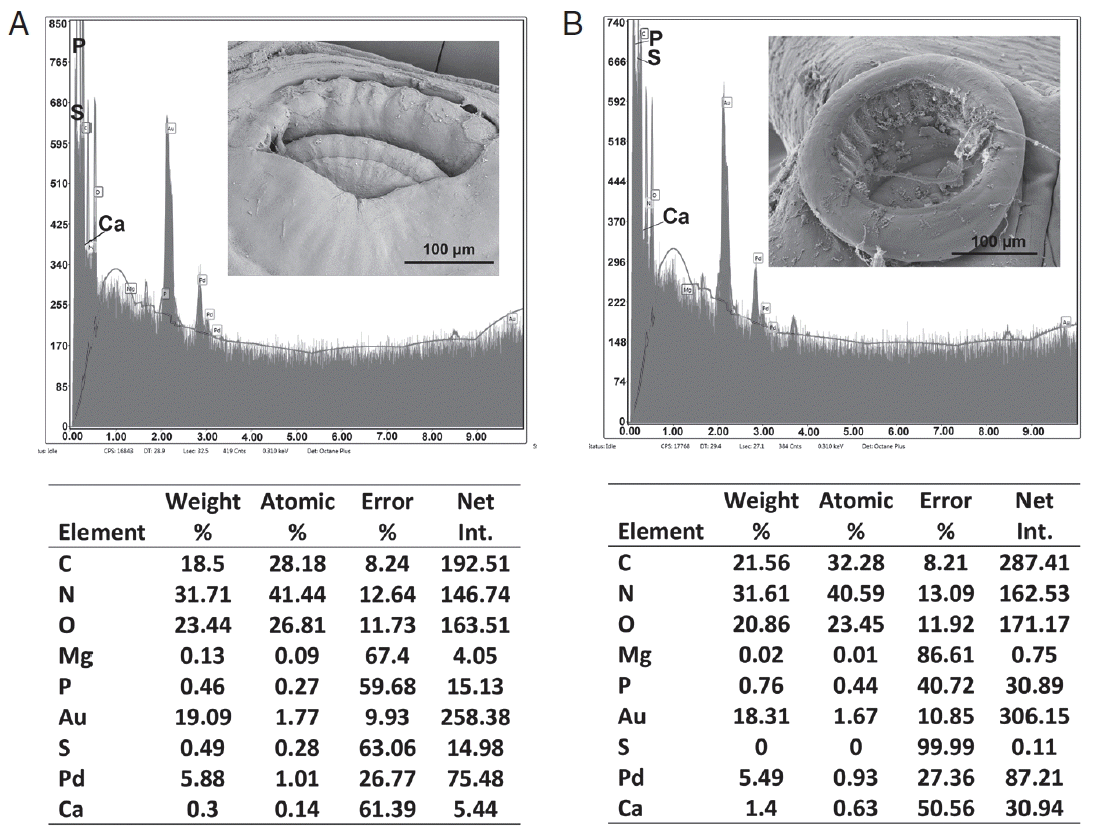
Structural analysis of different body parts of
O. hippopotami in both age groups are also reported for the first time and shows qualitative differences in the presence of hardening ions (S, P, Ca) in attachment structures (oral and haptor suckers) that increases with the age of the worm. EDXA has been used to track the change in chemical ions for areas of a parasite that become hardened with age (forming chemical bonds such as sulfide bonds for amino acids, apatite formation). This allows the parasite to harden certain areas of the body for attachment to the host. In the case of
Oculotrema, this tendency for suckers of haptor and in oral sucker was observed. The skeletal complex of the suckers of
Oculotrema is incredibly complex, consisting of few layers of 3 annular zones [
9], which provide a strong retention of the parasite, which is impeded by the constant movement of the eyelid throughout the parasite’s lifetime. Chemical compounds of these structures change with the age of the parasite, while its body grows 3–5 times bigger with a general tendency of accumulating of hardening ions (Ca, S, P) in the areas of the attachment structures. This tendency is very weak in body wall structure, where accumulation of hardening ions changes insignificantly with age (
Table 3). The same tendencies were shown for the hooks of acanthocephalans [
24–
26].