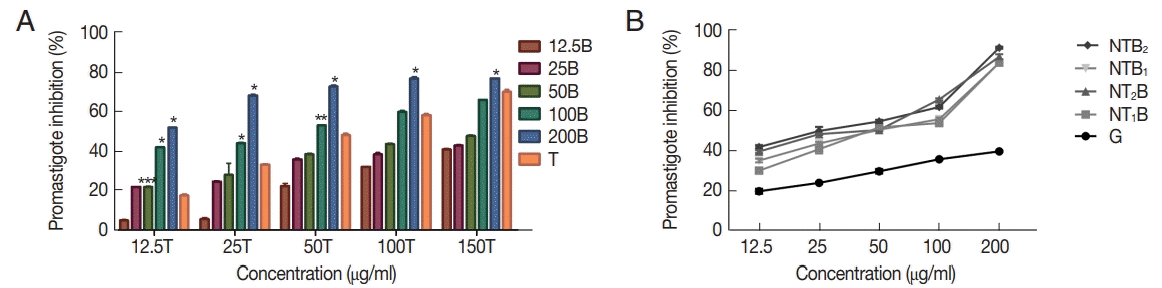
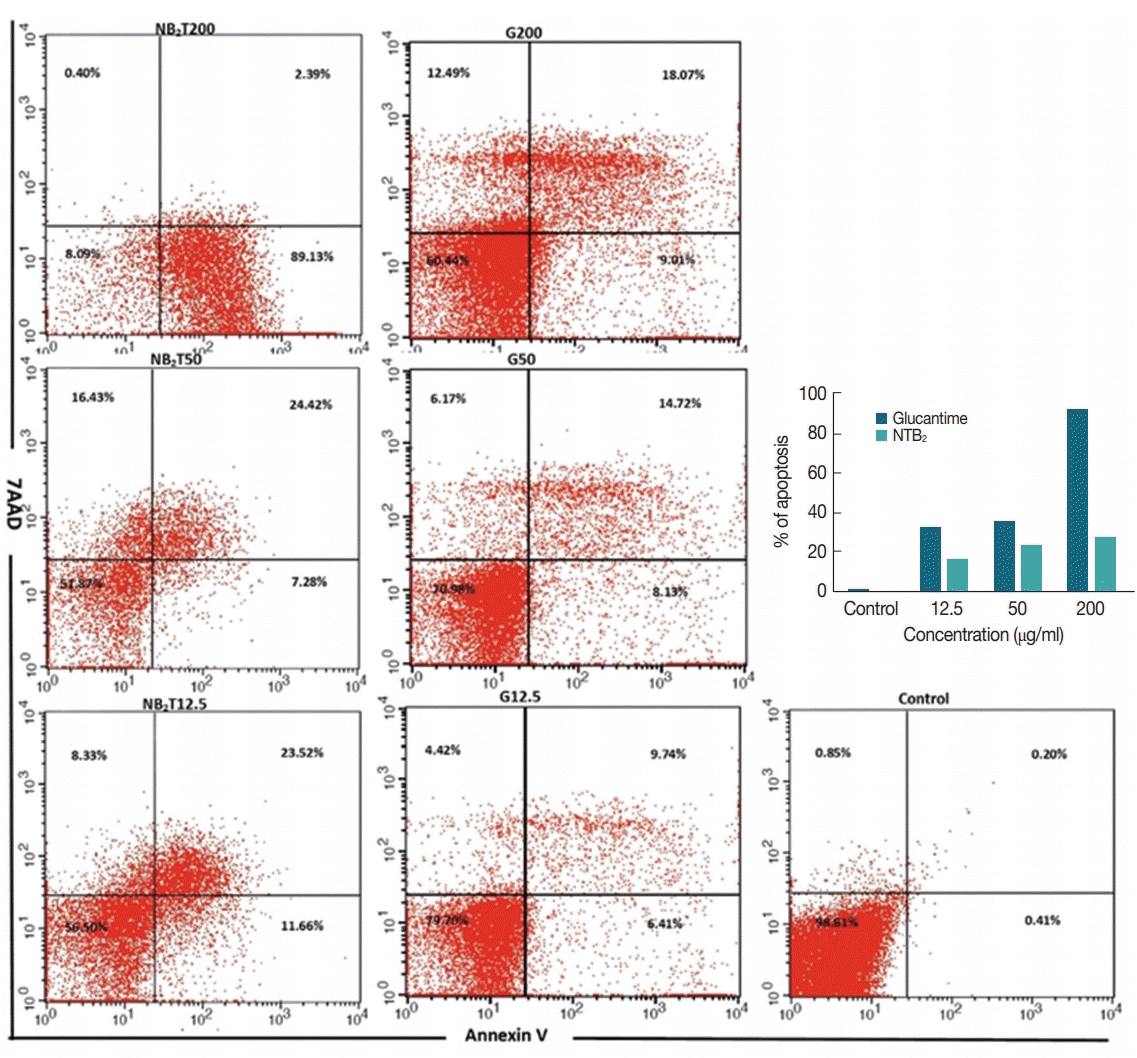
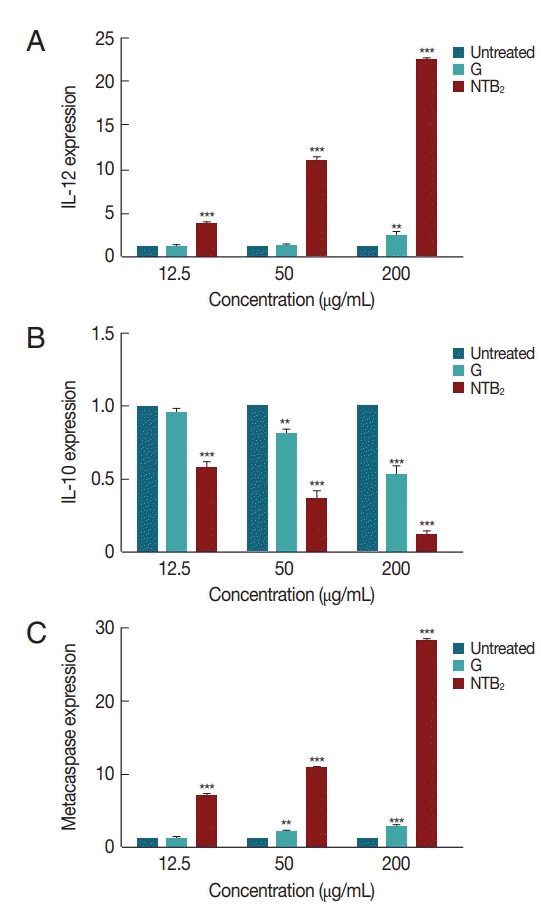
1. Cortez de Sá J, Almeida-Souza F, Mondêgo-Oliveira R, Oliveira Idos S, Lamarck L, Magalhães Ide F, Ataídes-Lima AF, Ferreira Hda S, Abreu-Silva AL. Leishmanicidal, cytotoxicity and wound healing potential of
Arrabidaea chica Verlot.
BMC Complement Altern Med 2016;16:1.



2. de Vries HJ, Reedijk SH, Schallig HD. Cutaneous leishmaniasis: recent developments in diagnosis and management.
Am J Clin Dermatol 2015;16:99-109.



3. Plano D, Baquedano Y, Moreno-Mateos D, Font M, Jiménez-Ruiz A, Palop JA, Sanmartín C. Selenocyanates and diselenides: a new class of potent antileishmanial agents.
Eur J Med Chem 2011;46:3315-3323.


4. Sharifi F, Sharifi I, Zarean M, Parizi MH, Aflatoonian M, Harandi MF, Zahmatkesh R, Mashayekhi M, Kermanizadeh A. Spatial distribution and molecular identification of
Leishmania species from endemic foci of south-eastern Iran.
Iran J Parasitol 2012;7:45-52.


5. Kazi KM, Mandal AS, Biswas N, Guha A, Chatterjee S, Behera M, Kuotsu K. Niosome: a future of targeted drug delivery systems.
J Adv Pharm Technol Res 2010;1:374-380.



6. Daie Parizi MH, Karvar M, Sharifi I, Bahrampour A, Heshmat Khah A, Rahnama Z, Baziar Z, Amiri R. The topical treatment of anthroponotic cutaneous leishmaniasis with the tincture of thioxolone plus benzoxonium chloride (Thio-Ben) along with cryotherapy: A single-blind randomized clinical trial.
Dermatol Ther 2015;28:140-146.


7. Kim JE, Kim HS, Shin YJ, Lee CS, Won CH, Lee SA, Lee JW, Kim YS, Kang JS, Ye SK, Chung MH. LYR71, a derivative of trimeric resveratrol, inhibits tumorigenesis by blocking STAT3-mediated matrix metalloproteinase 9 expression.
Exp Mol Med 2008;40:514-522.



8. Kim BH, Roh E, Lee HY, Lee IJ, Ahn B, Jung SH, Lee H, Han SB, Kim Y. Benzoxathiole derivative blocks lipopolysaccharide-induced nuclear factor-kappaB activation and nuclear factor-kappaB-regulated gene transcription through inactivating inhibitory kappaB kinase beta.
Mol Pharmacol 2008;73:1309-1318.


9. Kim BH, Min YS, Choi JS, Baeg GH, Kim Y, Shin JW, Kim TY, Ye SK. Benzoxathiol derivative BOT-4-one suppresses L540 lymphoma cell survival and proliferation via inhibition of JAK3/STAT3 signaling.
Exp Mol Med 2011;43:313-321.



10. Pardakhty A, Shakibaie M, Daneshvar H, Khamesipour A, Mohammadi-Khorsand T, Forootanfar H. Preparation and evaluation of niosomes containing autoclaved
Leishmania major: a preliminary study.
J Microencapsul 2012;29:219-224.


11. Attia IA, El-Gizawy SA, Fouda MA, Donia AM. Influence of a niosomal formulation on the oral bioavailability of acyclovir in rabbits.
AAPS Pharm Sci Tech 2007;8:206-212.

12. Shahiwala A, Misra A. Studies in topical application of niosomally entrapped Nimesulide.
J Pharm Pharm Sci 2002;5:220-225.

13. Chandra D, Naik S.
Leishmania donovani infection down-regulates TLR2-stimulated IL-12p40 and activates IL-10 in cells of macrophage/monocytic lineage by modulating MAPK pathways through a contact-dependent mechanism.
Clin Exp Immunol 2008;154:224-234.



14. Satheesh Kumar S, Gokulasuriyan RK, Ghosh M. Comparative in-silico genome analysis of
Leishmania (
Leishmania)
donovani: a step towards its species specificity.
Meta Gene 2014;2:782-798.



15. Yasinzai M, Khan M, Nadhman A, Shahnaz G. Drug resistance in leishmaniasis: current drug-delivery systems and future perspectives.
Future Med Chem 2013;5:1877-1888.


16. Rahimpour Y, Hamishehkar H. Niosomes as carrier in dermal drug delivery.
Recent Advances in Novel Drug Carrier Systems 2012;1:141-164.

17. Yasam VR, Jakki SL, Natarajan J, Kuppusamy G. A review on novel vesicular drug delivery: proniosomes.
Drug Deliv 2014;21:243-249.


18. Barcinski MA, DosReis GA. Apoptosis in parasites and parasite-induced apoptosis in the host immune system: a new approach to parasitic diseases.
Braz J Med Biol Res 1999;32:395-401.


19. Paris C, Loiseau PM, Bories C, Bréard J. Miltefosine induces apoptosis-like death in
Leishmania donovani promastigotes.
Antimicrob Agents Chemother 2004;48:852-859.



20. Das R, Roy A, Dutta N, Majumder HK. Reactive oxygen species and imbalance of calcium homeostasis contributes to curcumin induced programmed cell death in
Leishmania donovani
.
Apoptosis 2008;13:867-882.


21. Nguewa PA, Fuertes MA, Cepeda V, Iborra S, Carrión J, Valladares B, Alonso C, Pérez JM. Pentamidine is an antiparasitic and apoptotic drug that selectively modifies ubiquitin.
Chem Biodivers 2005;2:1387-1400.


22. Das M, Mukherjee SB, Shaha C. Hydrogen peroxide induces apoptosis-like death in
Leishmania donovani promastigotes.
J Cell Sci 2001;114:2461-2469.

23. Weingärtner A, Kemmer G, Müller FD, Zampieri RA, Gonzaga dos Santos M, Schiller J, Pomorski TG.
Leishmania promastigotes lack phosphatidylserine but bind annexin V upon permeabilization or miltefosine treatment.
PLoS One 2012;7:e42070.



24. Basmaciyan L, Azas N, Casanova M. Calcein+/PI- as an early apoptotic feature in
Leishmania
.
PLoS One 2017;12:e0187756.



25. Hingorani R, Deng J, Elia J, Mcintyre C, Mittar D. Detection of Apoptosis Using the BD Annexin V FITC Assay on the BD FACSVerse™ System. San Jose, USA. BD Biosciences 2011;1-12.
26. Mera-Ramírez A, Castillo A, Orobio Y, Gómez MA, Gallego-Marin C. Screening of TNFα, IL-10 and TLR4 single nucleotide polymorphisms in individuals with asymptomatic and chronic cutaneous leishmaniasis in Colombia: a pilot study.
BMC Infect Dis 2017;17:177.



27. Martorelli D, Muraro E, Merlo A, Turrini R, Fae DA, Rosato A, Dolcetti R. Exploiting the interplay between innate and adaptive immunity to improve immunotherapeutic strategies for Epstein-Barr-virus-driven disorders.
Clin Dev Immunol 2012;3:931952.

28. Bogdan C, Vodovotz Y, Nathan C. Macrophage deactivation by interleukln 10.
J Exp Med 1991;174:3-7.

29. Ambit A, Fasel N, Coombs GH, Mottram JC. An essential role for the
Leishmania major metacaspase in cell cycle progression.
Cell Death Differ 2008;15:113-122.


30. González IJ, Desponds C, Schaff C, Mottram JC, Fasel N.
Leishmania major metacaspase can replace yeast metacaspase in programmed cell death and has arginine-specific cysteine peptidase activity.
Int J Parasitol 2007;37:161-172.


31. Lee N, Gannavaram S, Selvapandiyan A, Debrabant A. Characterization of metacaspases with trypsin-like activity and their putative role in programmed cell death in the protozoan parasite
Leishmania
.
Eukaryot Cell 2007;6:1745-1757.



32. Debrabant A, Lee N, Bertholet S, Duncan R, Nakhasi HL. Programmed cell death in trypanosomatids and other unicellular organisms.
Int J Parasitol 2003;33:257-267.