INTRODUCTION
The majority of cryptosporidiosis in man and animals have been documented as intestinal cryptosporidiosis by
C. parvum. Infection with
C. muris in cattle may be asymptomatic or associated with transient symptom, therefore one has no concern for relating to the impact of the parasite on it's hosts. Recently,
C. muris was rediscovered in the abomasum of cattle that had failed to eat well and failed to gain body weight optimally (
Anderson, 1987). Thereafter, Esteban and Anderson (
1995) reported that cows shedding
C. muris oocysts produced significantly less milk (approximately 3.2 kg/day) and that mean daily milk production was significantly associated with shedding status of oocysts. The protozoon resides in the peptic glands of the abomasum, causing dilation of the glands and mucosal hypertrophy. Microvilli of gland epithelial cells are destroyed at attachment sites of the parasite. Plasma pepsinogen concentrations are twice as high in parasitized than in non-parasitized animals (
Anderson, 1987 &
1989).
Even though cows shedding
C. muris oocysts produced significantly less milk with decreased growth rate, no reports, so far, have been published on the kinetics in details of
C. muris infection in ruminants excepting fragmentary delineations (
Anderson, 1987 &
1988). It is reasonable to use calf as an experimental animal to study the basic relationship of low productivity in cattle attributed to the protozoon. However, calf is not suitable to feeding and maintaining due to its large size in addition to low establishment (success rate) of transmission experiment (
Anderson, 1988). Therefore, kids and lambs were selected as an animal model instead of calf. This has been examined by assessing the life cycle comprising of oocyst production and entity of developmental stages of the parasite in the abomasum of ruminants throughout the transmission experiments. The immunogenicity of the coccidium and oocyst reproduction were observed to elucidate the relationship between abomasal cryptosporidiosis and the protozoon.
MATERIALS AND METHODS
Animals used for the transmission experiments were Korean native kids and Corriedale lambs delivered from their dams free from oocyst discharge. One- to twenty-day-old nine kids and two lambs were inoculated per os with a single dose of 2 × 10
7 oocysts of
C. muris (strain MCR) originated from mice, as previously described (
Rhee et al., 1995). Oocysts have been maintained in our laboratory since 1990. Experimental animals were separately housed in wire-floored cages with concentrates, roughage and fresh water available
ad libitum. The cages were placed on trays containing 5 mm depth of water to keep the feces wet.
To determine the infectivity of the oocysts discharged from the kids to mice. 10 SPF ICR mice, 3-week-old, were each inoculated orally with a single dose of 2 × 106 oocysts originated from the kids.
While, three days after oocyst shedding ceased, 2 kids that had shed small numbers of oocysts were intramuscularly injected with 55 mg prednisolone acetate daily for 14 days to reproduce oocysts. After complete shedding of oocysts, a lamb and a kid that had shed a large number of oocysts were each challenged orally with a single dose of 2 × 107 oocysts generated from the lamb or the kid to observe the immunogenicity of the parasite. To compare morphological dimension of oocysts in relation to their origin, 50 oocysts from each origin were measured by an ocular micrometer.
Following inoculations, the procedures used for the feeding of the mice and the examination of fecal samples of the ruminants and mice were subjected to those described previously by Rhee et al. (
1995).
To observe the developmental stages of the parasite, a 2-week-old kid inoculated orally with a single dose of 2 × 107 oocysts was necropsied on day 44 postinoculation (PI), when discharged 6 × 107 oocysts/day. The abomasal segments about 0.5 cm in each length were removed from peptic glandular region (spiral fold), fixed in 10% neutral formalin, and submitted to routine histologic processing. Paraffin-embedded sections were stained with hematoxylin and eosin, and PAS stain. In addition, the abomasal segments were fixed in Karnousky fixative (pH 7.4). The specimens were then washed in 0.1 M cacodylate buffer, postfixed in 2% sodium tetroxide solution in cacodylate buffer, and dehydrated in an ascending ethanol series. They were embedded in epoxy resin and sectioned with a diamond knife on a ultramicrotome. The ultrathin sections were stained with 20% uranyl acetate and examined with a JEM 1010 transmission electron microscope.
DISCUSSION
Although the contention for taxonomy is continued until current, C. muris and C. parvum are valid, and the organism discovered in the abomasum of cattle is C. muris because ultrastructure of developmental stages, location of parasitism and the prepatent and patent periods differ from each other enormously.
Age-dependent resistance to
Cryptosporidium spp. or susceptibility to cryptosporidiosis of animals varies with the species of protozoa as well as. age of the host. Lambs became resistant to experimental infection with
C. parvum isolated from calves by about 15-20 days of age, and those infected at 30 days of age became infected with the organism and shed oocysts for a brief periods (1-2 days), but did not develop clinical signs of disease or growth retardation (
Tzipori et al,, 1981). Studies of
C. parvum infection in several strains of laboratory mice have demonstrated that 4- to 6-day-old mice were more susceptible to experimental infection than those of 21 or 42 days old (
Sherwood et al, 1982;
Heine et al., 1984). Sreter et al (
1995) indicated that both innate and acquired resistances to
C. baileyi were age-related in chickens.
On the other hand, we corroborated that susceptibilities to experimental infection with
C. muris (strain MCR) in mice at 3, 12 and 15 wk of age were similar (Rhee et al., unpublished observations).
Cryptosporidium muris appears to be associated with mild diarrhea in cattle of all ages, especially young animals (
Upton and Current, 1985). Anderson (
1988) described that abomasal cryptosporidiosis was produced in only one of 18 calves exposed 2 hr pre-clostrally to millions of fresh oocysts without presenting data in details. Given the data presented here, although it is well known that younger animals are more susceptible to
Cryptosporidium infection, susceptibility to
C. muris infection in kids and lambs may be irrespective of age like mice, because of a low relationship between age and intensity of oocyst outputs. However, we could not conclude age-independent resistance to
C. muris in kids and lambs because the number of experimental animals are not sufficient.
Infection with
C. muris appears to be chronic in cattle: the pattern of shedding oocysts in the steers was continuous over a period of 4 months and as of his writing, he had possession of three cattle which had shed
C. muris oocyst for over a year at the rate of about one million per gram of feces (
Anderson, 1987 &
1989). However, the prepatent and patent periods of the parasite for ruminants have not been established throughout transmission experiment as yet.
Our previous work (
Rhee et al., 1995) revealed that a patent period ranged from 61 to 64 days with a prepatent period of 9-11 days for mice inoculated orally with various doses of
C. muris (strain MCR) oocyst. While, Iseki et al. (
1989) intimated that the prepatent and patent periods of
C. muris (strain RN 66) for mice, guinea pigs, rabbits, dogs and cats were exceedingly different from each other. Based on the present findings, the prepatent and patent periods for kids and lambs were longer than those reported for various laboratory animals by Iseki et al. (
1989) and Rhee et al (
1995).
The significant differences in patency and the total oocyst outputs were noted depending on individual hosts. The standard deviations of the means of patency and the total oocyst outputs were high (47.8 ± 21.1 days and 156.6 × 10
6 ± 253.6 × 10
6), whereas the lengths of the prepatent period were similar to one another. Those of the prepatent period was relatively low (28.1 ± 5.8 days), while there was no correlation between the length of the patent period and the total oocyst output, indicating a significant individual difference in terms of the effectiveness of the acquired resistance to
C. muris (
Table 1). Therefore, even by same strain of inoculum, the prepatent and patent periods of
C. muris vary significantly with the species of host.
In the present study, no oocysts were excreted from a lamb and a kid by challenge infections at any time, which implies that the immunogenicity of the parasite is very strong like mice. Moreover, we ascertained the infectivity of the oocysts discharged from the kids to mice and reappearance of latent Cryptosporidia after primary infection by prednisolone acetate administration like mice. However, it is concluded that ruminants such as cattle, goat and sheep are not suitable hosts for the protozoon because infection with greater numbers of oocyst output was not found to occur with a familar findings in ruminants.
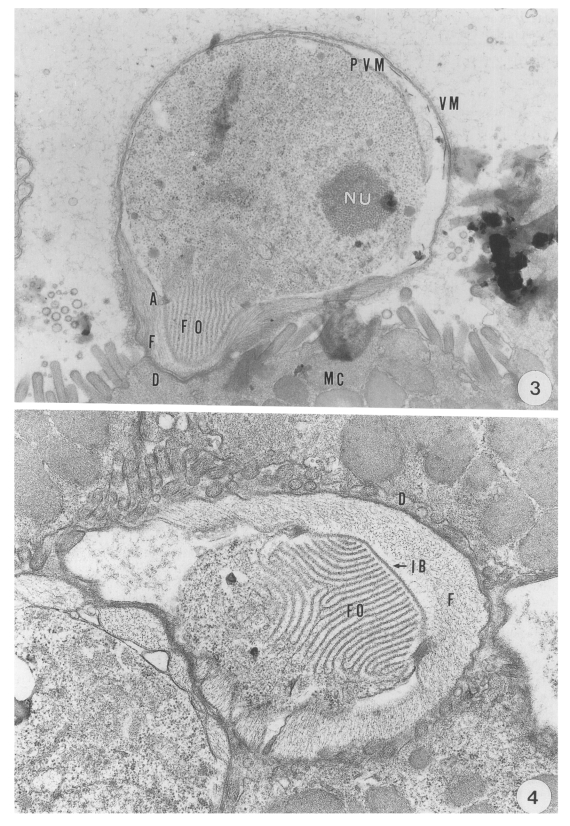
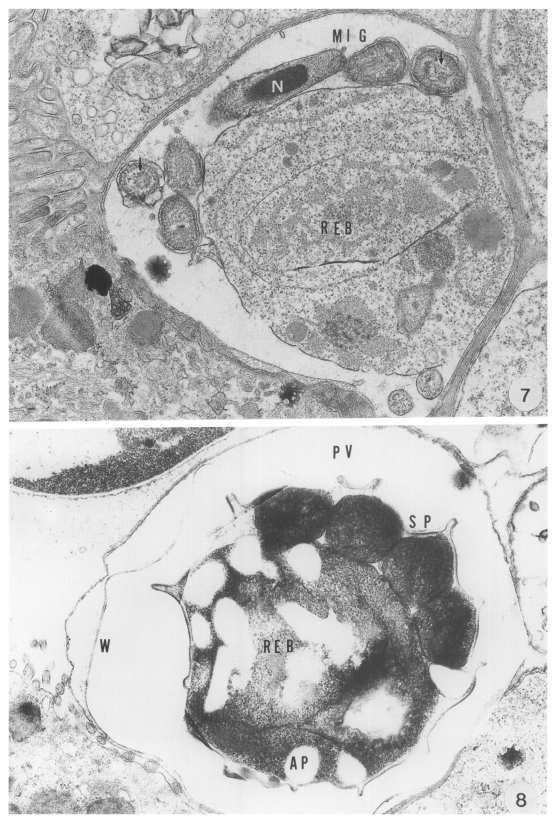
The present study primarily documented that the ultrastructural features of the attachment site of the parasite to the mucus cells differed remarkably from those of
C. parvum in the abomasum of ruminants. A unique structure of the anterior projection of the parasite was seen identical with that observed in the stomach of mice, as previously described by Uni et al. (
1987) and Rhee et al (
1991b). Moreover, the size of the parasite was greater at each developmental stage than that of
C. parvum. In addition, massive populations of this cryptosporidia were seen in the abomasal peptic glands, most of which were found infected with them.
Therefore, the present paper clarified the full course of the host-parasite interaction in ruminants throughout the transmission experiment and ultrastructure of the developmental stages with confirming greater numbers of cryptosporidia in the gastric peptic glands and fecal samples of kids and lambs.