Abstract
Sparganosis is a parasitic zoonosis caused by plerocercoid larvae of Spirometra species, commonly transmitted through ingestion of infected copepods or raw intermediate hosts. A 51-year-old man presented with a palpable mass in his right thigh. Surgical excision revealed a worm-like structure. Histopathological and serologic findings suggested sparganosis, and PCR amplification of the cox1 gene from paraffin-embedded tissue showed 99% sequence identity with Spirometra erinaceieuropaei (GenBank accession No. KJ599680.1). Praziquantel (75 mg/kg/day for 3 days) was administered, and the patient showed no evidence of recurrence during follow-up. Eosinophilia was not observed; however, positive sparganum antibodies supported the diagnosis. This case demonstrates that molecular identification using mitochondrial genes can be a valuable diagnostic tool, especially when morphological features are limited. Furthermore, it highlights the zoonotic potential of S. erinaceieuropaei and underscores the importance of food safety, hygiene education, and continuous epidemiological surveillance to prevent human sparganosis in endemic regions.
-
Key words: Spirometra erinaceieuropaei, human sparganosis, cox1 gene, phylogenetic analysis, Korea
Introduction
Sparganosis is a parasitic infection caused by the plerocercoid larvae of the genus
Spirometra [
1]. The genus
Spirometra includes over 60 identified tapeworm species, notably
Spirometra erinaceieuropaei (syn.
S. erinacei),
S. mansoni,
S. mansonoides, and
S. decipiens with more than 2,000 human infections reported globally [
2].
Human sparganosis occurs through ingestion of water contaminated with infected crustaceans or by consuming second intermediate hosts [
2]. The first intermediate hosts are typically freshwater copepods in which the procercoid larvae develop, and when these are consumed by fish, reptiles, or amphibians that serve as the second intermediate hosts, the procercoids subsequently mature into plerocercoid larvae [
3]. Definitive hosts, such as dogs and cats, become infected by ingesting the second intermediate hosts, and the adult worms eventually develop in the small intestine of these animals [
2]. Additionally, some species of
Spirometra can utilize paratenic hosts, where the larvae remain infective without further development [
4].
Sparganosis is a zoonotic disease that occurs sporadically in humans. Although cases have been reported worldwide, the majority have been documented in Asia [
5]. When humans become infected, the larvae typically migrate to subcutaneous tissues, forming cyst-like nodules that may cause discomfort, hemiparesis, seizures, headaches, and peripheral eosinophilia [
6]. Here, we report a human case of
S. erinaceieuropaei infection identified by molecular analysis in Korea.
Case Report
A 51-year-old man presented with a palpable mass on his right thigh, first noticed in February 2024. In April 2024, he underwent an ultrasound examination at an outside hospital, which revealed a 3-cm-sized mass. On April 17, surgical excision of the subcutaneous benign tumor was performed. A worm-like structure was discovered, and the specimen was submitted for histopathological examination. On May 22, 2024, the patient visited the Infectious Diseases Department at Pusan National University Yangsan Hospital for further evaluation and a second opinion. The physical examination revealed no additional palpable lesions, and chest radiography showed no remarkable findings. His past medical history was negative for diabetes mellitus, hypertension, viral hepatitis, and tuberculosis. He had not been on any chronic medications; however, he had independently taken albendazole once weekly for 3 consecutive weeks after suspecting a parasitic infection. He denied consuming raw or undercooked meat but reported frequently eating raw vegetables. No additional clinical abnormalities were observed apart from the thigh lesion. Serologic testing for parasitic antibodies was subsequently performed. Complete blood count showed: WBC 6,050/mm³, hemoglobin 15.0 g/dl, platelets 254,000/mm³, absolute neutrophil count 3,160/mm³, and eosinophil count 140/mm³. There was no evidence of eosinophilia. Clinically, the findings were consistent with sparganosis, and serological tests were positive for sparganum antibodies. The patient was treated with praziquantel 4.8 g/day (75 mg/kg/day) for 3 consecutive days. No recurrence was observed following treatment, and follow-up was unremarkable. Written informed consent was obtained from the patient for publication of this case report, including clinical data, pathological images, and genetic information.
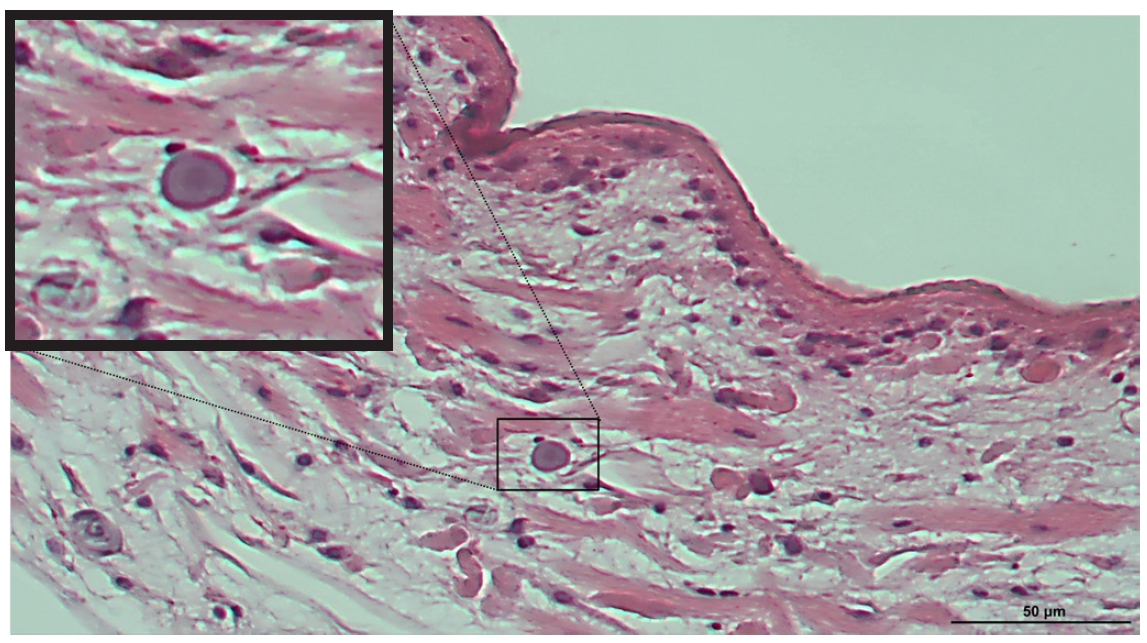
The parasite recovered during surgery was immediately fixed in formalin and embedded in paraffin for histopathological analysis. The transverse section showed a characteristic pattern of noncellular tegument, cellular subtegument, and parenchyma containing calcareous corpuscles are visible (
Fig. 1). To identify the specific species, DNA was extracted from the paraffin-embedded tissue, and primers were designed targeting the
cox1 gene. The primers p1f (5´-TGG TTT TTT GGA CAT CCT GAA-3´) and p1r (5´-ATC ACA TAA TGA AAG TGA GCC-3´) amplified a 440 bp product corresponding to positions 707–1,146 of the
cox1 gene [
7]. The PCR product showed 99% sequence identity with the mitochondrial genome of
S. erinaceieuropaei (GenBank accession No. KJ599680.1). This sequence was submitted to the GenBank database (accession No. PX415209).
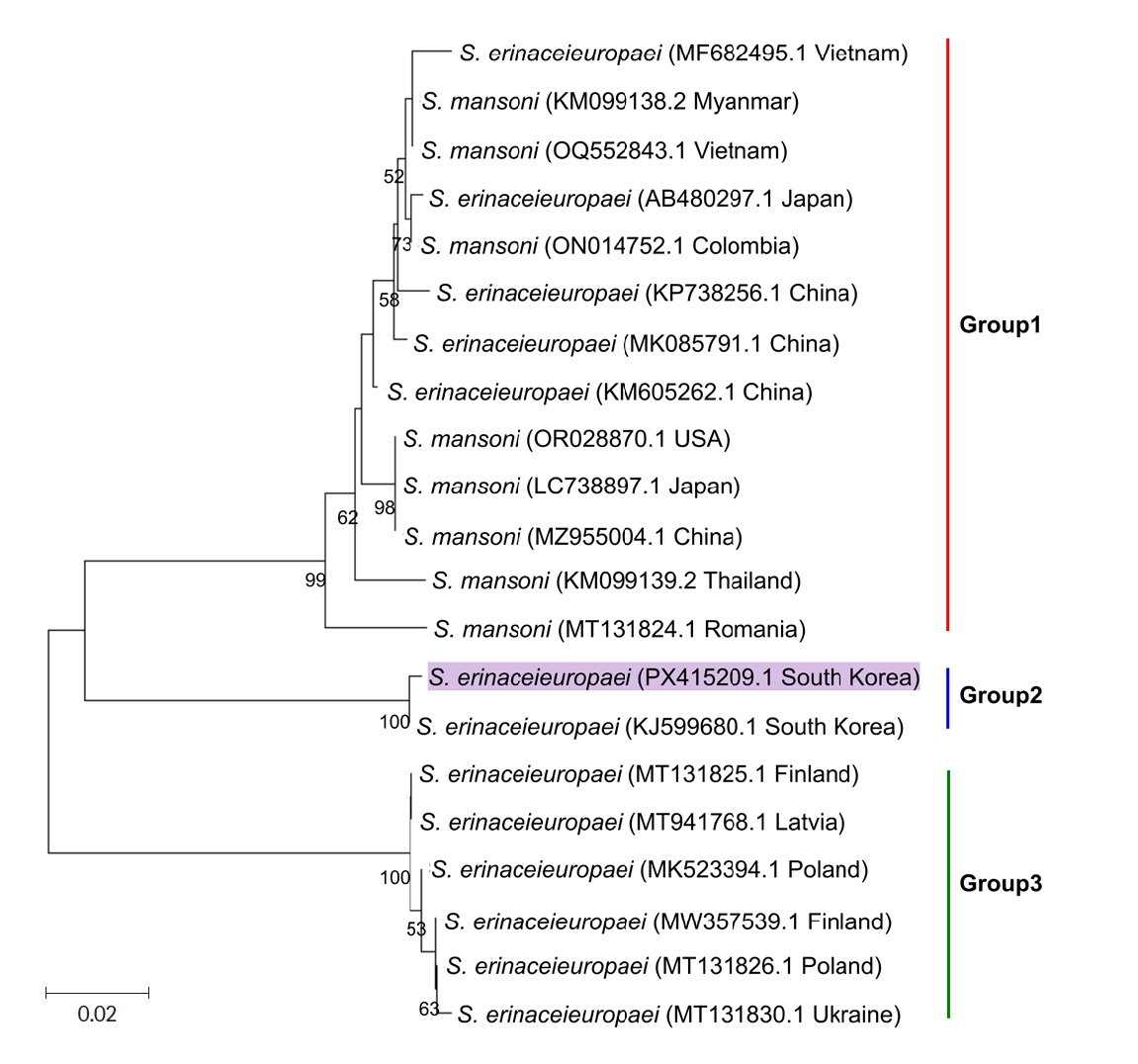
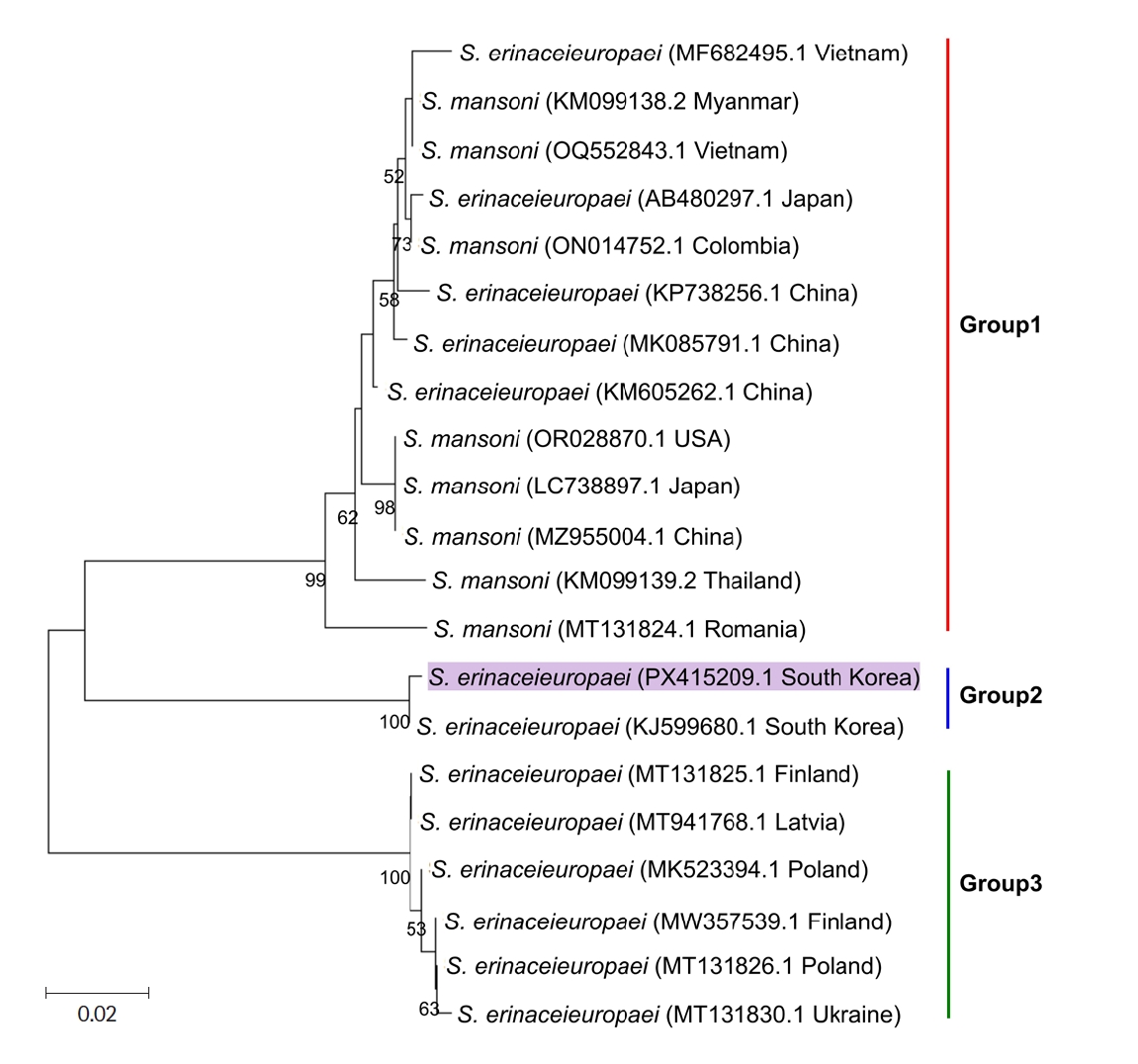
To investigate global genetic similarity, we constructed a phylogenetic tree comparing
cox1 sequences of
S. erinaceieuropaei from various countries across Europe, Asia, and the Americas (
Fig. 2). Phylogenetic analysis based on 21 global
cox1 sequences revealed 3 distinct clades, and the 2 Korean isolates formed an exclusive cluster (Group 2), separate from Asian, European, and American strains. This indicates a unique genetic lineage among Korean
S. erinaceieuropaei isolates. The phylogenetic tree was constructed using the MEGA11 software program, which is available at
https://www.megasoftware.net.
Discussion
Human infection with
Spirometra tapeworms can cause severe tissue damage or even death when larvae migrate to various organs and tissues [
8]. Sparganosis has been reported worldwide, with approximately 400 cases documented in Korea alone [
3]. The clinical manifestations vary depending on the final location of the sparganum. Subcutaneous sparganosis typically presents as nodular lesions beneath the skin [
9]. Infected patients may exhibit symptoms such as cough, headache, dizziness, fatigue, fever, and a sense of movement at the infection site [
6,
10]. Although sparganosis remains a relatively rare parasitic disease, most patients present with mass-like lesions or nonspecific symptoms. Surgical removal is generally regarded as the primary treatment for sparganosis. Although antiparasitic agents such as praziquantel have been used adjunctively in selected cases, their therapeutic efficacy remains unclear [
11].
In Korea, infections caused by
S. erinaceieuropaei are most frequently reported [
3]. In contrast, both
S. erinaceieuropaei and
S. mansoni are equally prevalent in other regions [
12]. In the present case,
S. erinaceieuropaei was identified. We amplified the
cox1 gene of
S. erinaceieuropaei using specifically designed primers, and the obtained genomic DNA sequence matched
S. erinaceieuropaei. Calcareous corpuscles are characteristic structures of cestode larvae and serve as an important diagnostic marker for identifying
Spirometra spp. In the present case, these structures were clearly observed within the larval tissue (
Fig. 1).
Phylogenetic analysis was performed using MEGA11, incorporating the sequence from this case along with 20 related sequences from NCBI. Sequences reported from other Asian regions, Europe, and the United States belonged to distinct groups, demonstrating clear genetic differentiation. These findings indicate that
S. erinaceieuropaei isolated in Korea is genetically closely related to Asian sequences, suggesting the existence of genetic diversity associated with geographic distribution. Such phylogenetic variation may reflect not only regional genetic differentiation but also potential differences in pathogenicity and host specificity that could influence clinical manifestations [
13]. Therefore, future molecular epidemiological studies integrating sequence data from a wider range of geographic regions and host species are warranted to elucidate how genetic divergence among groups affects pathogenicity, transmission dynamics, and clinical outcomes.
In conclusion, this study reports a molecularly identified case of S. erinaceieuropaei infection in Korea and emphasizes the need for increased awareness of sparganosis in endemic areas. Furthermore, this study supports the use of PCR-based and sequence-based molecular diagnostic methods are essential for accurate species identification and epidemiological investigations. Considering the zoonotic nature of this disease, public health interventions such as promoting safe dietary habits, improving hygiene, and community education should be strongly encouraged.
Notes
-
Author contributions
Conceptualization: Choi SY. Data curation: Park MK. Formal analysis: Choi SY. Investigation: Choi SY, Jeong YJ. Methodology: Jung BK, Lim SJ, Yu HS. Resources: Lim SJ. Supervision: Yu HS. Visualization: Choi SY. Writing – original draft: Choi SY. Writing – review & editing: Jung BK, Lim SJ, Yu HS.
-
Conflict of interest
Hak Sun Yu serves as an editor of Parasites, Hosts and Diseases but had no involvement in the decision to publish this article. No other potential conflicts of interest relevant to this study were reported.
Fig. 1.Histopathological findings of Spirometra erinaceieuropaei in thigh tissue of the present case. The recovered parasite was fixed in formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Characteristic basophilic calcareous corpuscles were clearly observed within the parasite and are also shown in the magnified view. Scale bar=50 μm
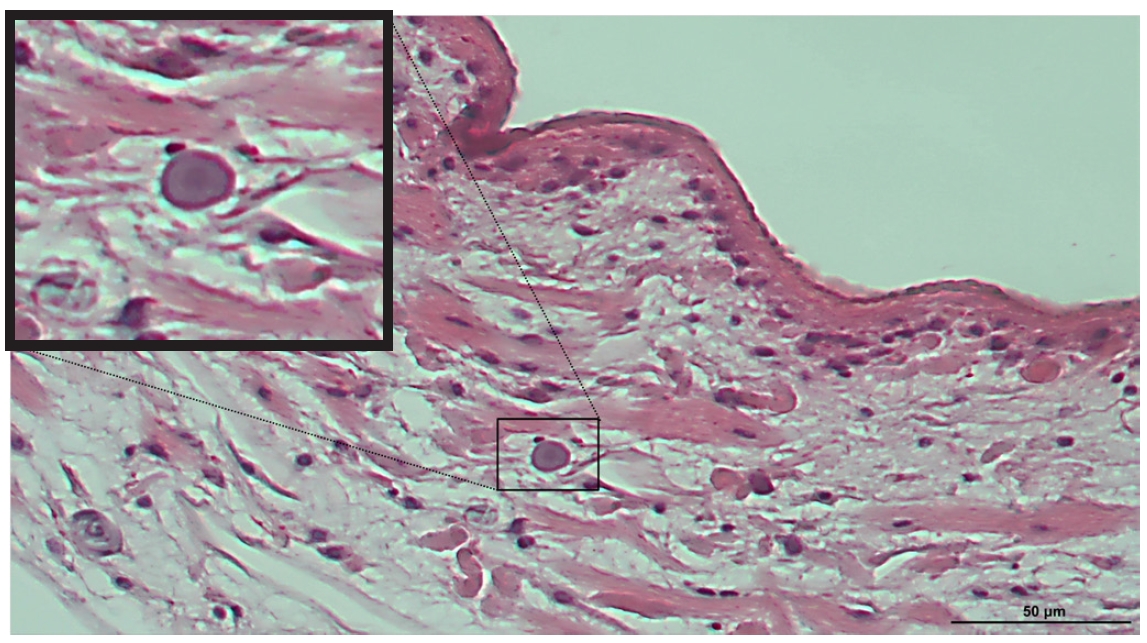
Fig. 2.Phylogenetic analysis of Spirometra spp. A neighbor-joining tree was constructed using the sequence obtained in this study along with 13 sequences of S. erinaceieuropaei and 8 of S. mansoni retrieved from the NCBI GenBank database. Numbers at the nodes represent bootstrap values based on 1,000 replicates.
References
- 1. Tokiwa T, Fushimi M, Chou S, et al. Aberrant sparganosis in cat caused by Spirometra mansoni (Cestoda: Diphyllobothriidae): a case report. BMC Vet Res 2024;20:148. https://doi.org/10.1186/s12917-024-03995-z
- 2. Tappe D, Berger L, Haeupler A, et al. Case report: molecular diagnosis of subcutaneous Spirometra erinaceieuropaei sparganosis in a Japanese immigrant. Am J Trop Med Hyg 2013;88:198-202. https://doi.org/10.4269/ajtmh.2012.12-0406
- 3. Kim JG, Ahn CS, Sohn WM, Nawa Y, Kong Y. Human sparganosis in Korea. J Korean Med Sci 2018;33:e273. https://doi.org/10.3346/jkms.2018.33.e273
- 4. Kondzior E, Kowalczyk R, Tokarska M, et al. Multispecies reservoir of Spirometra erinaceieuropaei (Cestoda: Diphyllobothridae) in carnivore communities in north-eastern Poland. Parasit Vectors 2020;13:560. https://doi.org/10.1186/s13071-020-04431-5
- 5. Wiwanitkit V. A review of human sparganosis in Thailand. Int J Infect Dis 2005;9:312-6. https://doi.org/10.1016/j.ijid.2004.08.003
- 6. Liu Q, Li MW, Wang ZD, Zhao GH, Zhu XQ. Human sparganosis, a neglected food borne zoonosis. Lancet Infect Dis 2015;15:1226-35. https://doi.org/10.1016/S1473-3099(15)00133-4
- 7. Jeon HK, Park H, Lee D, et al. Human infections with Spirometra decipiens plerocercoids identified by morphologic and genetic analyses in Korea. Korean J Parasitol 2015;53:299-305. https://doi.org/10.3347/kjp.2015.53.3.299
- 8. Liu W, Gong T, Chen S, et al. Epidemiology, diagnosis, and prevention of sparganosis in Asia. Animals (Basel) 2022;12:1578. https://doi.org/10.3390/ani12121578
- 9. Hwang JM, Hwang DS, Kang C, Lee JW. Subcutaneous sparganosis mimicking soft tissue tumor: a case report. Int Med Case Rep J 2019;12:47-50. https://doi.org/10.2147/imcrj.S192764
- 10. Lee KJ, Myung NH, Park HW. A case of sparganosis in the leg. Korean J Parasitol 2010;48:309-12. https://doi.org/10.3347/kjp.2010.48.4.309
- 11. Le AT, Do LT, Nguyen HT, Nguyen HT, Do AN. Case report: the first case of human infection by adult of Spirometra erinaceieuropaei in Vietnam. BMC Infect Dis 2017;17:669. https://doi.org/10.1186/s12879-017-2786-x
- 12. Kuchta R, Phillips AJ, Scholz T. Diversity and biology of Spirometra tapeworms (Cestoda: Diphyllobothriidea), zoonotic parasites of wildlife: a review. Int J Parasitol Parasites Wildl 2024;24:100947. https://doi.org/10.1016/j.ijppaw.2024.100947
- 13. Kumar A, Singh KP, Bali P, et al. iNOS polymorphism modulates iNOS/NO expression via impaired antioxidant and ROS content in P. vivax and P. falciparum infection. Redox Biol 2018;15:192-206. https://doi.org/10.1016/j.redox.2017.12.005
Citations
Citations to this article as recorded by

 , Mi0 -Kyung Park1
, Mi0 -Kyung Park1 , Yu Jin Jeong1
, Yu Jin Jeong1 , Bong-Kwang Jung1
, Bong-Kwang Jung1 , Seungjin Lim2,3,*,†
, Seungjin Lim2,3,*,† , Hak Sun Yu1,2,*
, Hak Sun Yu1,2,*