Abstract
The egg morphology of minute intestinal flukes (MIF) that can occur as human infections in the Republic of Korea, i.e., Metagonimus yokogawai, M. miyatai, M. takahashii, Heterophyes nocens, Heterophyopsis continua, Stellantchasmus falcatus, Stictodora fuscata, Pygidiopsis summa, and Gymnophalloides seoi, was studied in comparison with Clonorchis sinensis. The adult worms were obtained from residents of endemic areas, and their intrauterine eggs were studied and measured using light microscopy; the length, width, length-width ratio (LWR), and Faust-Meleney index (FMI). Several specimens were processed for scanning electron microscopy (SEM), and before gold-coating, the uterine portion of each fluke was etched with a sharp pin in order to expose the eggs. The MIF eggs were ovoid, pyriform, or elliptical with a size range of 21-35×12-21 µm. S. fuscata eggs revealed the highest FMI (largest in the area) and lowest LWR, whereas P. summa eggs showed the lowest FMI and medium LWR. SEM revealed that G. seoi and S. fuscata had remarkably clean shell surface lacking the muskmelon-like structure which is prominent in C. sinensis eggs. In Metagonimus spp., H. continua, H. nocens, and S. falcatus eggs, minute surface ridges were recognizable though less prominent compared with C. sinensis. On the surface of P. summa eggs, thread-like curly structures were characteristically seen. The results revealed that important differential keys for MIF eggs include the length, width, area (FMI), shape of the eggs, and the extent of the muskmelon-like structure or ridges on their shell surface and operculum.
-
Key words: Metagonimus yokogawai, Metagonimus miyatai, Metagonimus takahashii, Heterophyes nocens, Heterophyopsis continua, Stellantchasmus falcatus, Stictodora fuscata, Pygidiopsis summa, Gymnophalloides seoi, Clonorchis sinensis, egg, trematode, ultrastructure
INTRODUCTION
At least 70 species of foodborne intestinal trematodes are known to occur as human infections around the world [
1]. In the Republic of Korea, about 20 species are known, which consist of species belonging to the family Heterophyidae (=heterophyids), Echinostomatidae, Gymnophallidae (=gymnophallids), Plagiorchiidae, and Neodiplostomidae [
2]. Among them, the heterophyid and gymnophallid flukes are minute in their body size, so they are often called as minute intestinal flukes (MIF) [
3]. Their eggs are also minute and morphologically close to one another, and it is practically difficult to differentiate the species when they are discharged in the human feces unless they are apparently smaller or larger than others or extremely unique in shape.
The zoonotic MIF species that reproduce small-sized eggs so far known in the Republic of Korea include 11 species, namely,
Metagonimus yokogawai,
Metagonimus miyatai,
Metagonimus takahashii,
Heterophyes nocens,
Heterophyopsis continua,
Stellantchasmus falcatus,
Stictodora fuscata,
Stictodora lari,
Pygidiopsis summa,
Gymnophalloides seoi, and
Acanthotrema felis [
2,
4]. The eggs of these flukes are 18-35 µm in length and 12-19 µm in width. In our previous study, the uterine eggs of
M. yokogawai,
H. nocens,
H. continua,
S. falcatus, and
P. summa were taken out and examined in comparison with those of
C. sinensis using light microscopy [
5]. The study concluded that the 6 kinds of eggs could be morphologically differed if careful observation and measurement of sufficient numbers of eggs were done [
5]. Nevertheless, specific diagnosis has been difficult in areas of no previously known endemicity as well as in areas with mixed infections with different species of heterophyids [
6]. In such areas, recovery of adult flukes is needed to obtain a definite diagnosis [
6]. However, to provide tentative diagnosis of MIF infections, additional information is needed on the comparative morphology of MIF eggs. In the present study, we examined the egg morphology of 9 MIF species by means of light microscopy and scanning electron microscopy (SEM).
MATERIALS AND METHODS
Collection of worms
The following 9 MIF species were studied; M. yokogawai (Katsurada, 1912), M. takahashii Suzuki, 1930, M. miyatai Saito et al., 1997, P. summa Onji and Nishio, 1916, H. nocens Onji and Nishio, 1915, H. continua (Onji and Nishio, 1916), S. falcatus Onji and Nishio, 1916, S. fuscata (Onji and Nishio, 1916), and G. seoi Lee, Chai and Hong, 1993. The adult specimens of these flukes were obtained from people who live in endemic areas of various localities (Shinan-gun, Gangjin-gun, Umseong-gun, Aphae-do, and Jeju-do in the Republic of Korea) after treatment with praziquantel and purging with MgSO4. Adult specimens of C. sinensis, which were used for controls, were obtained from a human patient and also from an experimental rat infected with metacercariae from the freshwater fish.
Measurements of egg size
The worms obtained from stool samples were washed with PBS several times and fixed in 10% formalin. Some worms were stained with acetocarmine, and others were kept unstained. They were morphologically identified in both formalin-fixed and acetocarmine-stained specimens. Uterine eggs were examined and measured in formalin-fixed specimens under cover slip pressure using a light microscope, and pictures were taken. In each MIF species, the length and width of 10 uterine eggs were measured, and the mean±SD values were obtained. For better understanding of the egg dimensions, the length-width ratio (LWR=L/W) and the Faust-Meleney Index (FMI) (=L[length]×W
2 [width] of the eggs) [
7], were also calculated.
For SEM, the formalin-preserved worms were washed with 0.1 M phosphate buffer and post-fixed with 1% osmium tetroxide for 2 hr at 4℃. The fixed worms were dehydrated in graded series of ethanol, and several worms were pooled for each species. The prepared worms were dried using a critical point dryer (CO2). They were mounted on stubs, and their body was dissected with a pin to etch the upper part of uterine branches and expose their eggs. Then, the surface of the worms and eggs was coated with gold. The surface ultrastructure of the uterine eggs was observed with a ABT DS-130C Scanning Electron Microscope (Tokyo, Japan). The eggs were photographed with a fixed magnification for all different MIF species and C. sinensis.
RESULTS
Egg size
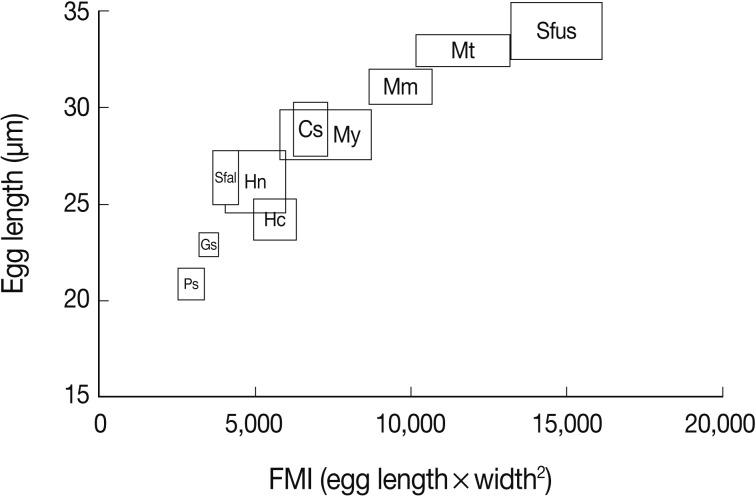
The length, width, and LWR of the eggs were variable by different MIF species (
Table 1;
Figs. 1-
30) and seemed to be helpful for specific diagnosis. In addition, FMI appeared to be another useful index for distinguishing the MIF eggs under light microscopy, although there were some overlappings between different species (
Table 1;
Fig. 31).
Based on the measurements, the eggs of
S. fuscata were the largest in length (33.6±1.4), width (20.8±0.7), and FMI (14,608±1,495), whereas the eggs of
P. summa were the smallest of all in length (20.9±0.9), width (12.1±1.0), and FMI (3,046±417) (
Table 1). With regard to the length, the second largest was
M. takahashii (32.6±0.8), followed by
M. miyatai,
M. yokogawai,
S. falcatus,
H. nocens,
H. continua, and
G. seoi (
Table 1). The length of
C. sinensis eggs (28.5±1.3) was almost the same as that of
M. yokogawai eggs (28.4±1.3). In the egg width, the second widest was
M. takahashii (18.9±1.3), followd by
M. miyatai,
M. yokogawai,
H. continua,
H. nocens,
S. falcatus, and
G. seoi (
Table 1). The width of
C. sinensis eggs (15.7±1.0) was almost the same as that of
H. continua eggs (15.5±1.2) and slightly smaller than that of
M. yokogawai eggs (16.1±1.4). The LWR was the largest for
S. falcatus (2.1±0.1) and the smallest for
S. fuscata (1.6±0.1). In FMI values, the second largest was
M. takahashii (11,646±1,492), followed by
M. miyatai,
M. yokogawai,
H. continua,
H. nocens,
S. falcatus, and
G. seoi (
Table 1). The FMI of
C. sinensis eggs (6,872±593) was greater than that of
H. continua (5,747±757) and
H. nocens (5,151±975) but smaller than that of
M. yokogawai (7,380±1,494) (
Table 1).
The adult flukes and their eggs were photographed under light microscopy and SEM (
Figs. 1-
30). In general, the eggs were ovoid, pyriform, or elliptical with a size range of 21-35×12-21 µm.
Metagonimus spp.,
H. nocens, and
H. continua eggs were ovoid, whereas
G. seoi eggs were elliptical.
C. sinensis,
P. summa,
S. falcatus, and
S. fuscata eggs were pyriform. However, the morphology of some eggs was very similar to one another.
P. summa eggs were highly similar to
C. sinensis eggs except in their smaller size. Eggs of 3
Metagonimus species were indistinguishable except in their comparative sizes; largest for
M. takahashii, followed by
M. miyatai and then
M. yokogawai. Eggs of
H. continua were similar to eggs of
H. nocens but the former was wider than the latter and stout. Eggs of
S. falcatus were slender and long compared with those of
H. nocens and
H. continua. Eggs of
S. fuscata were big and wide compared with those of
C. sinensis and
Metagonimus spp.
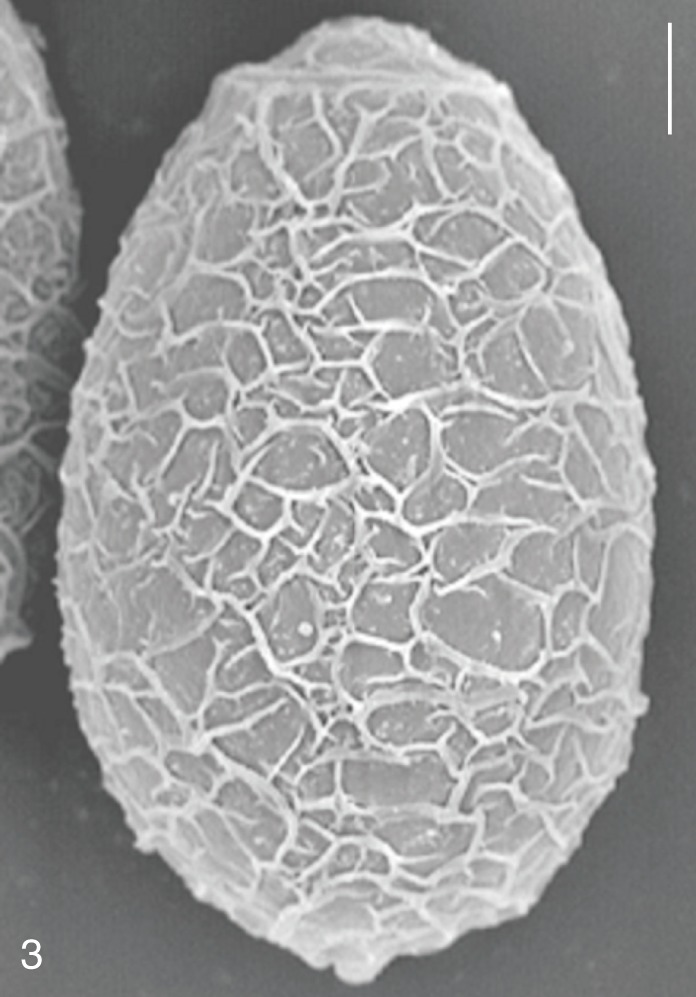
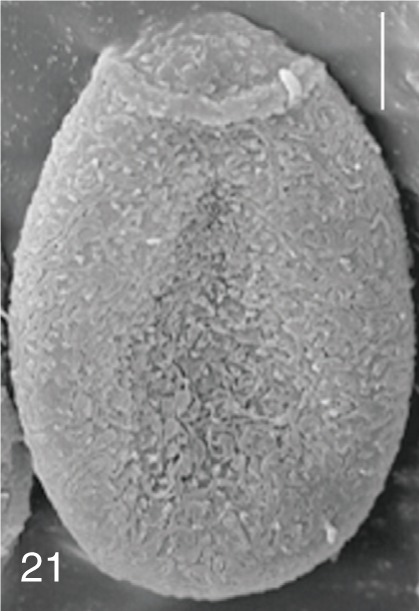
The surface ultrastructure of the MIF eggs appeared to be a good marker for differential diagnosis from
C. sinensis eggs. Whereas the surface of
C. sinensis eggs was covered with prominent, elevated ridges called muskmelon-like structures (
Fig. 3), these structures were not observed in all MIF species eggs studied (
Figs. 6,
9,
12,
15,
18,
21,
24,
27,
30). A similar surface morphology with
C. sinensis was seen in
P. summa eggs, in which thread-like curly structures were present (
Fig. 9). In 3
Metagonimus spp.,
H. continua,
H. nocens, and
S. falcatus eggs, multiple surface ridges were recognized (
Figs. 12,
15,
18,
21,
24,
27). However, the extent of the ridges was minute in
Metagonimus spp., and relatively conspicuous in
H. continua,
H. nocens, and
S. falcatus. By comparison, the egg shell surface was clean in
G. seoi and
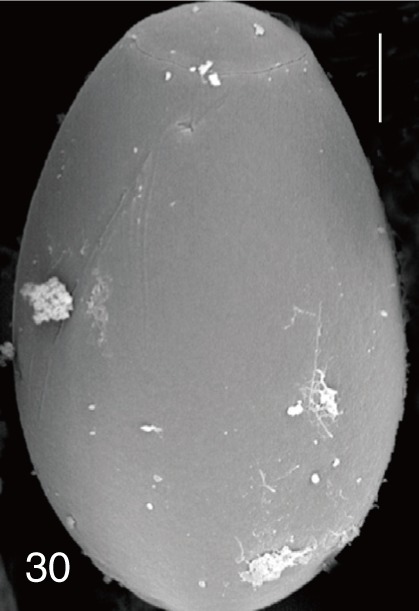
S. fuscata eggs (
Figs. 6,
30).
In some species, including
C. sinensis (
Fig. 3),
P. summa (
Fig. 9),
H. continua (
Fig. 21), and
H. nocens (
Fig. 24), the opercular junction was distinct and shoulder rims were prominent. However, shoulder rims were negligible in
G. seoi (
Fig. 6),
M. yokogawai (
Fig. 12), and
S. fuscata (
Fig. 30) eggs. The operculum was wide and big in
G. seoi (
Fig. 6), whereas it was narrow and small in
H. nocens (
Fig. 24) and
S. falcatus (
Fig. 27). The abopercular knob was present in some specimens of
C. sinensis,
P. summa, and
Metagonimus spp., but absent in others (
Figs. 3,
9,
15,
18).
DISCUSSION
MIF that include heterophyids and a gymnophallid represent significant health problems in various localities of the Republic of Korea [
2,
4,
6]. The diagnosis is done by fecal examinations. However, morphological discrimination of the eggs in the feces using light microscopy is problematic [
6]. The results of the present study revealed that not only the length, width, LWR, FMI, and shape of the eggs but also their surface ultrastructures as shown by SEM are distinctive markers that may be useful for discrimination of MIF eggs. The morphology of the shoulder rims, operculum, and abopercular knob was also helpful for the species differentiation.
The results of the present study were in good agreement with our previous study using light microscopy [
5]. However, in the present study, eggs of
G. seoi,
M. miyatai,
M. takahashii, and
S. fuscata were newly added for study, and significant additional findings were obtained. The surface ultrastructures of MIF eggs occurring in the Republic of Korea were seldom studied by SEM, with the exception of a recent paleoparasitological report on
C. sinensis,
M. yokogawai, and
G. seoi eggs [
8]. In other countries, several reports were published on the SEM morphology of
C. sinensis,
M. yokogawai,
M. takahashii (under the name
Metagonimus sp.), and
S. falcatus eggs [
9-
11]. The results of the present study on these trematode species were well corresponded with those of the previous reports [
8-
11].
In our study, the eggshell surface of
P. summa,
M. miyatai,
H. nocens,
H. continua, and
S. fuscata was studied for the first time using SEM. The surface ultrastructure of
P. summa eggs were unique, having thread-like curly structures, which appeared to be useful for differential diagnosis of eggs. The eggshell surface of 3
Metagonimus spp. was characterized by the presence of minute ridges, although these ridges were more prominent in
H. nocens,
H. continua, and
S. falcatus eggs. The present findings for
S. falcatus were similar to those reported previously [
11]. The surface ultrastructure of
S. fuscata eggs was very smooth which was similar to
G. seoi eggs [
8]. In light microscopy, the eggshells of
S. fuscata eggs were smooth and thick with an inconspicuous operculum [
4]. In our study,
S. fuscata eggs were much larger than those of
G. seoi and had a small operculum.
In Thailand and Lao PDR, differentiation of
Opisthorchis viverrini eggs from MIF eggs, including
Haplorchis taichui,
Haplorchis pumilio,
Haplorchis yokogawai,
Prosthodendrium molenkampi,
Phaneropsolus bonnei, and
S. falcatus was problematic, and studies on this subject were performed [
11-
14]. Light microscopic and SEM findings of these MIF eggs revealed unique features; for example,
Haplorchis spp. eggs were characterized by the presence of filamentous mesh-like structures or thread-like ridges on the shell surface [
11,
12]. In
O. viverrini eggs, the presence of muskmelon-like structures was characteristic and distinctly different from MIF eggs [
11-
13]. On the other hand, the presence of iodophilic bodies was characteristic in the eggs of
P. bonnei and
P. molenkampi, whereas such bodies were not seen in
O. viverrini eggs [
13,
14]. Use of potassium permanganate solution (1% w/v) in fecal examinations was also useful for differentiation of
O. viverrini eggs from
P. molenkampi and
P. bonnei eggs [
15].
In the present study, the FMI appeared to be a useful index for MIF egg differentiation. Although there were some overlappings in the range, the mean values of FMI were quite specific for each different species of fluke. This was in agreement with a previous report in which FMI was applied to differentiate MIF,
O. viverrini, and
C. sinensis eggs [
11].
ACKNOWLEDGMENT
The authors have no conflict of interest in relation with this report.
References
- 1. Chai JY. In Murrell KD, Fried B, editors. Intestinal Flukes. Food-borne Parasitic Zoonoses: Fish and Plant-borne Parasites. World Class Parasites. 2007. Vol. 11:New York, USA. Springer; p. 53-115.
- 2. Chai JY, Lee SH. Foodborne intestinal trematode infections in the Republic of Korea. Parasitol Int 2002;51:129-154.
- 3. Chai JY, Murrell KD, Lymbery AJ. Fish-borne parasitic zoonoses: Status and issues. Int J Parasitol 2005;35:1233-1254.
- 4. Cho SH, Cho PY, Lee DM, Kim TS, Kim IS, Hwang EJ, Na BK, Sohn WM. Epidemiological survey on the infection of intestinal flukes in residents of Muan-gun, Jeollanam-do, the Republic of Korea. Korean J Parasitol 2010;48:133-138.
- 5. Lee SH, Hwang SW, Chai JY, Seo BS. Comparative morphology of eggs of heterophyids and Clonorchis sinensis causing human infections in Korea. Korean J Parasitol 1984;22:171-180.
- 6. Chai JY, Shin EH, Lee SH, Rim HJ. Foodborne intestinal flukes in Southeast Asia. Korean J Parasitol 2009;47(suppl):S69-S102.
- 7. Faust EC, Meleney HE. Studies on schistosomiasis japonica. Am J Hyg 1924;Monogr Ser no. 3:1-339.
- 8. Shin DH, Lim DS, Choi KJ, Oh CS, Kim MJ, Lee IS, Kim SB, Shin JE, Bok GD, Chai JY, Seo M. Scanning electron microscope study of ancient parasite eggs recovered from Korean mummies of the Joseon Dynasty. J Parasitol 2009;95:137-145.
- 9. Ishii Y. Scanning electron microscopical study on the structure of parasite eggs. Fukuoka Acta Med 1971;63:419-431.
- 10. Ditrich O, Giboda M, Šerba J. Species determination of eggs of opisthorchiid and heterophyid flukes using scanning electron microscopy. Angew Parasitol 1990;31:3-9.
- 11. Ditrich O, Giboda M, Scholz T, Beer SA. Comparative morphology of eggs of the Haplorchiinae (Trematoda: Heterophyidae) and some other medically important heterophyid and opisthorchiid flukes. Folia Parasitol (Praha) 1992;39:123-132.
- 12. Kaewkes S, Elkins DB, Sithithaworn P, Haswell-Elkins MR. Comparative studies on the morphology of the eggs of Opisthorchis viverrini and lecithodendriid trematodes. Southeast Asian J Trop Med Public Health 1991;22:623-630.
- 13. Tesana S, Srisawangwonk T, Kaewkes S, Sithithaworn P, Kanla P, Arunyanart C. Eggshell morphology of the small eggs of human trematodes in Thailand. Southeast Asian J Trop Med Public Health 1991;22:631-636.
- 14. Sripa B, Bethony JM, Sithithaworn P, Kaewkes S, Mairiang E, Loukas A, Mulvenna J, Laha T, Hotez PJ, Brindley PJ. Opisthorchiasis and Opisthorchis-associated cholagiocarcinoma in Thailand and Laos. Acta Trop 2011;120(Suppl):S158-S168.
- 15. Sukontason K, Piangjai S, Sukontason K, Chaithong U. Potassium permanganate staining for differentiation the surface morphology of Opisthorchis viverrini, Haplorchis taichui and Phaneropsolus bonnei eggs. Southeast Asian J Trop Med Public Health 1999;30:371-374.
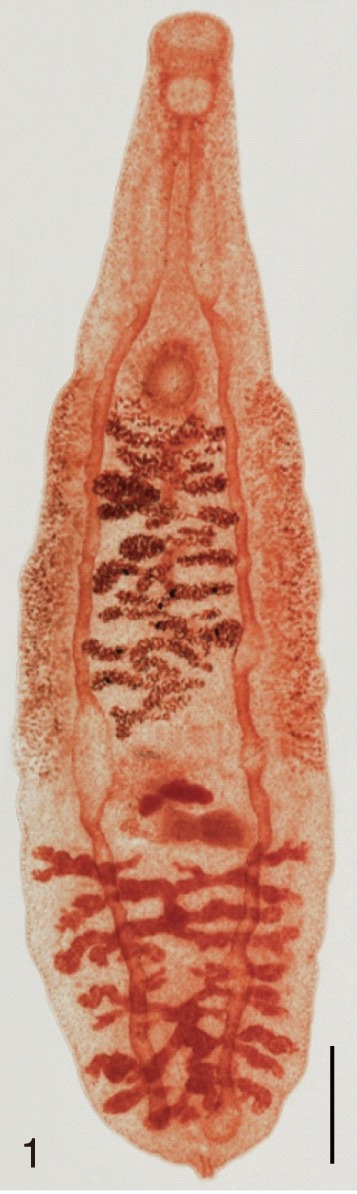
Fig. 1
Clonorchis sinensis. An adult worm obtained from an experimental rat. Bar=200 µm.
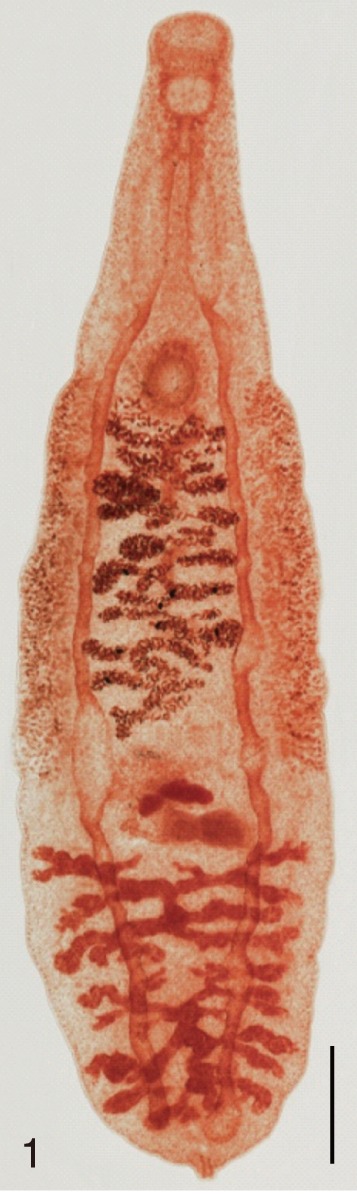
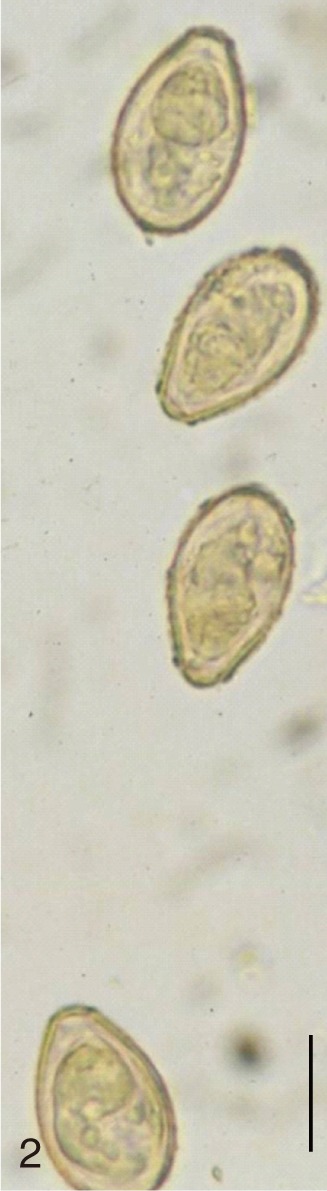
Fig. 2
Clonorchis sinensis. Uterine eggs freed and seen under a light microscope. Bar=15 µm.
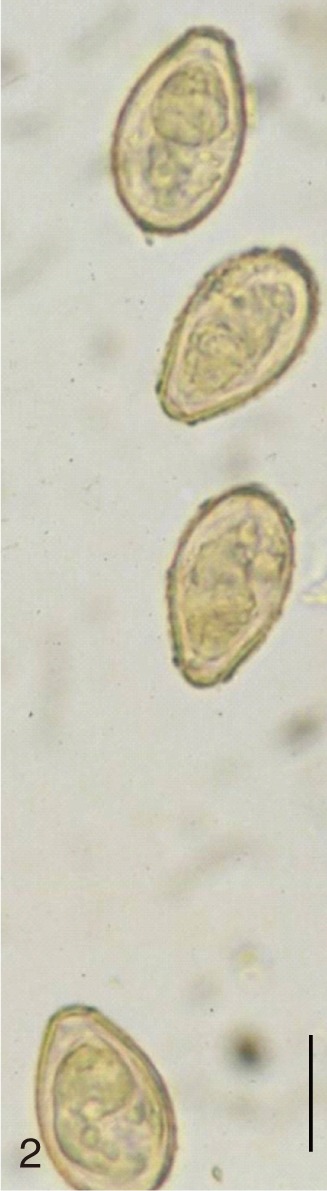
Fig. 3
Clonorchis sinensis. SEM view of an egg showing the typical muskmelon-like structure on the surface. Bar=3 µm.
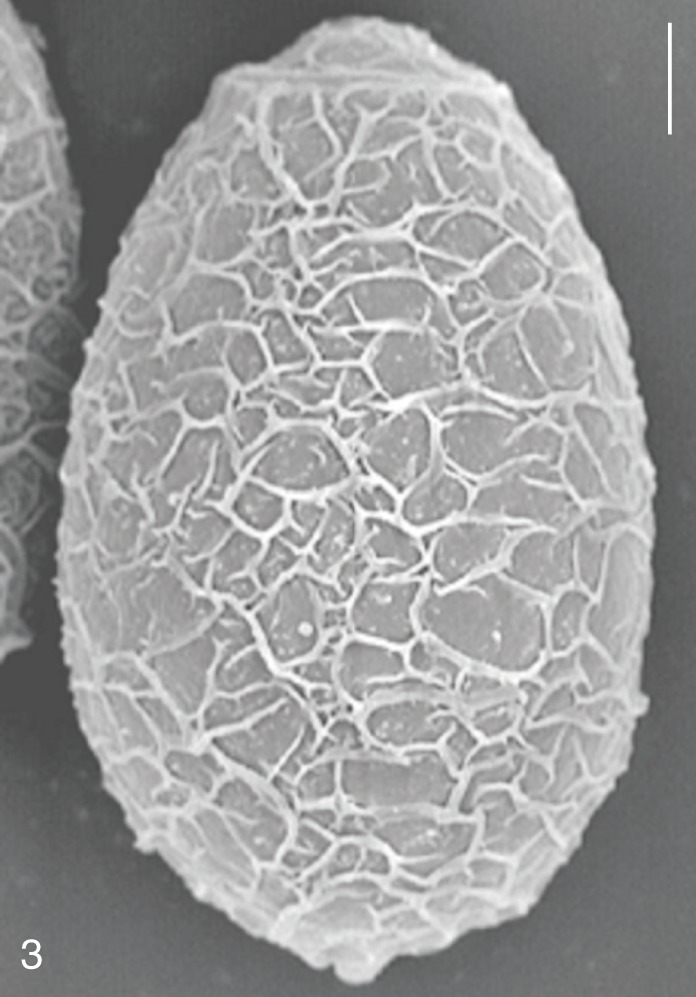
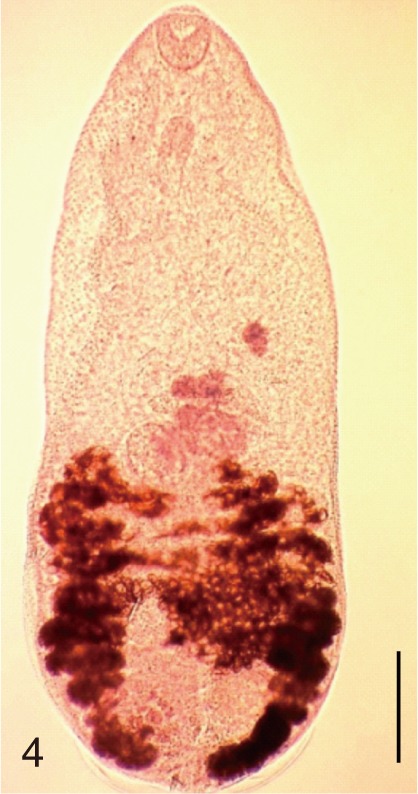
Fig. 4
Pygidiopsis summa. An adult worm obtained from a naturally infected person on Aphae-do, Korea. Bar=100 µm.

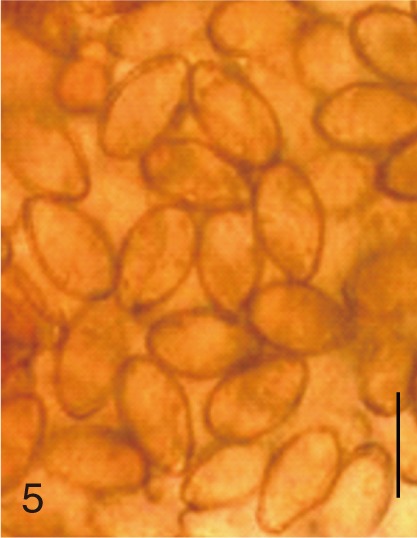
Fig. 5
Pygidiopsis summa. Uterine eggs. Bar=20 µm.
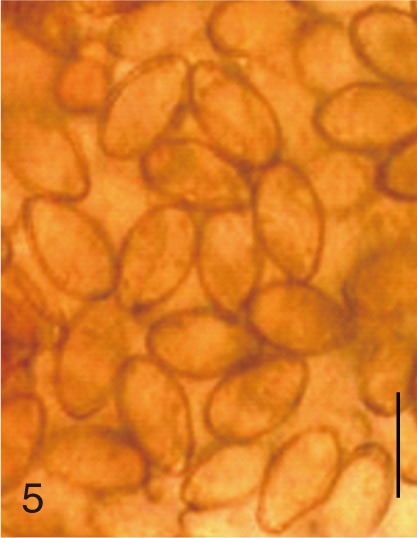
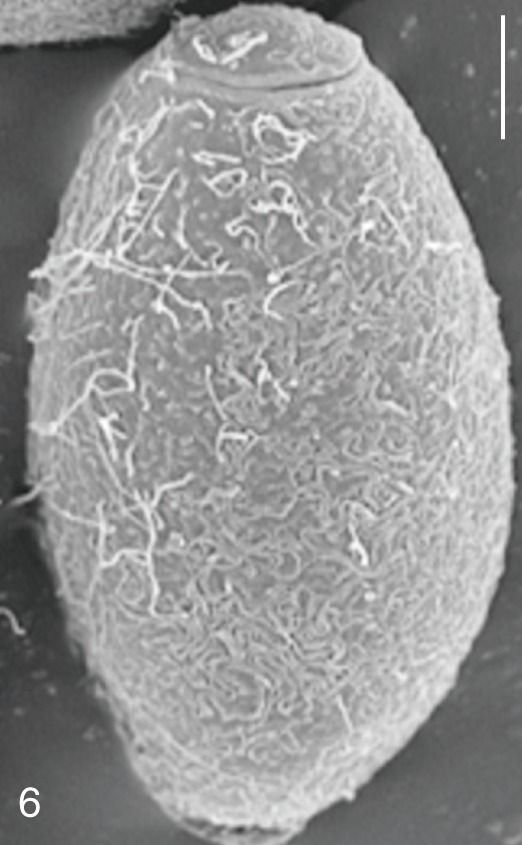
Fig. 6
Pygidiopsis summa. SEM view of an egg showing the thread-like ridges on the surface. Bar=3 µm.
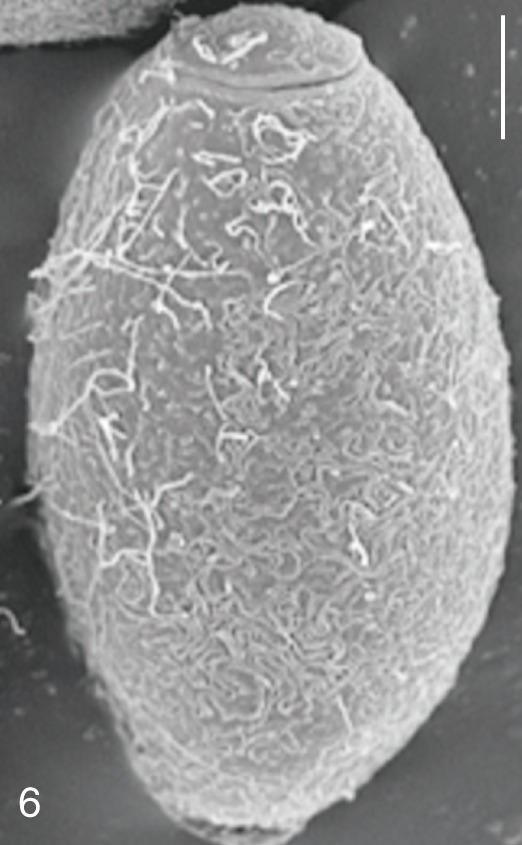
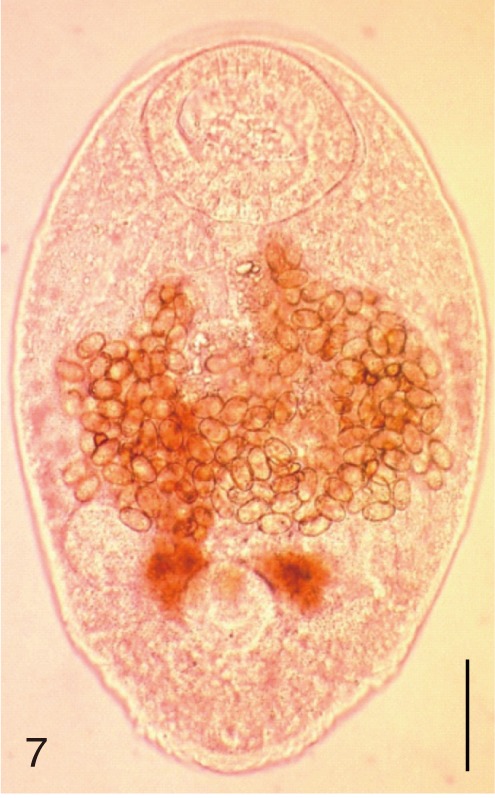
Fig. 7
Gymnophalloides seoi. An adult worm obtained from a naturally infected person on Aphae-do, Korea. Bar=80 µm.
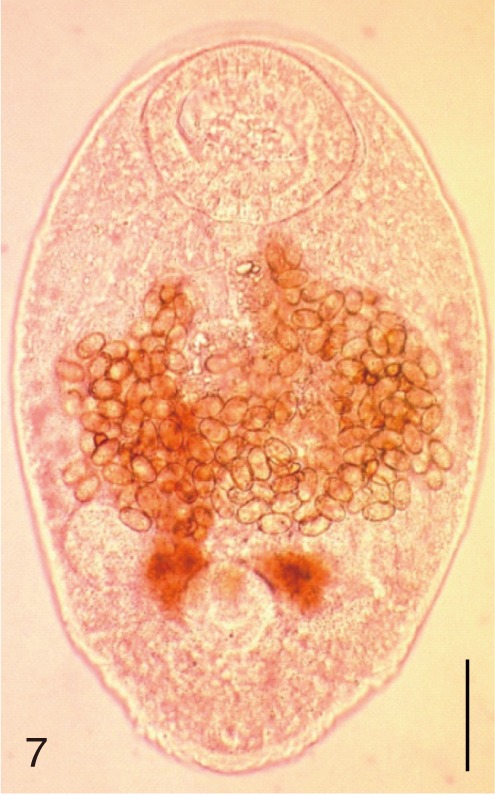
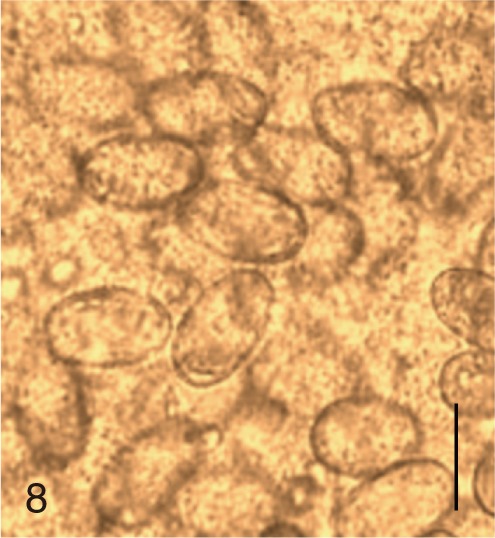
Fig. 8
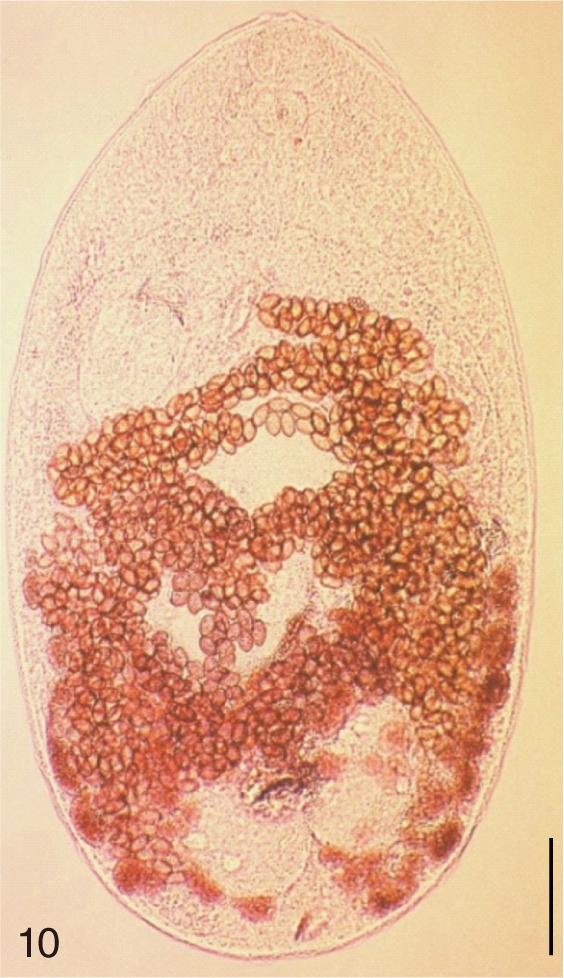
Gymnophalloides seoi. Uterine eggs. Bar=20 µm.
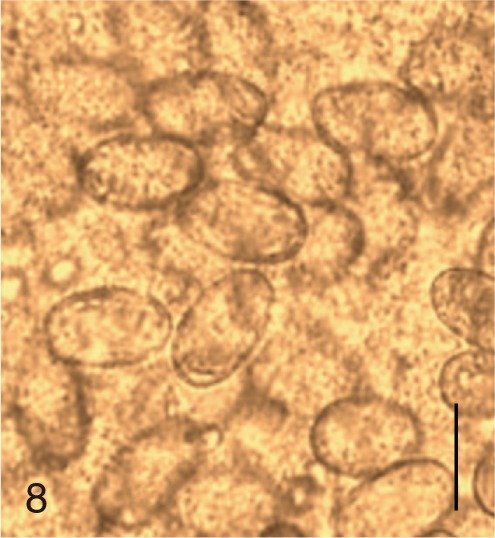
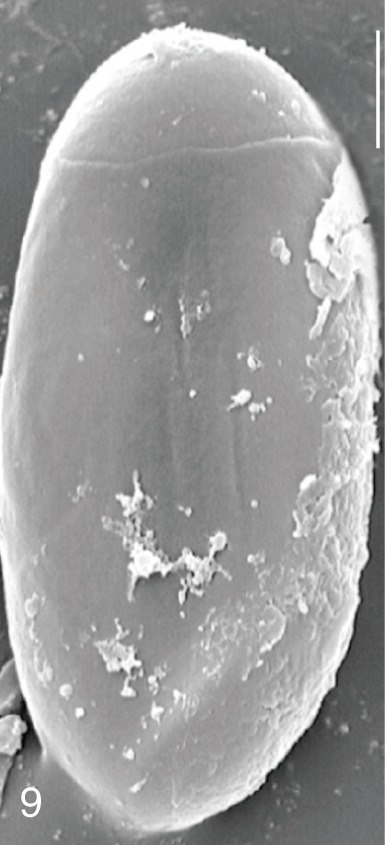
Fig. 9
Gymnophalloides seoi. SEM view of an egg showing a big operculum and clean shell surface. Bar=3 µm.
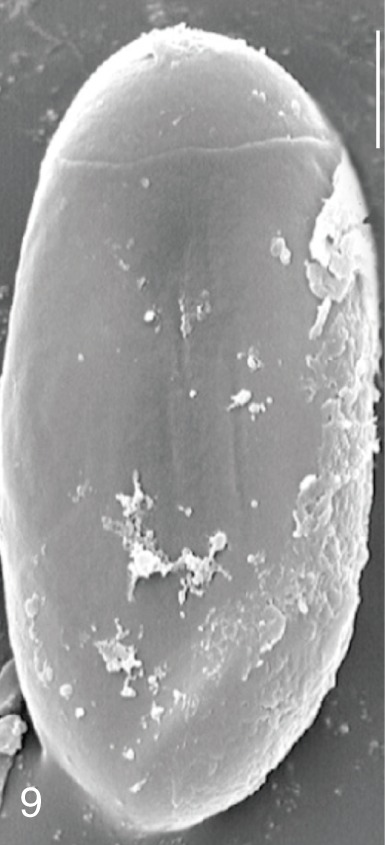
Fig. 10
Metagonimus yokogawai. An adult worm obtained from a naturally infected person on Jeju Island, Korea. Bar=150 µm.
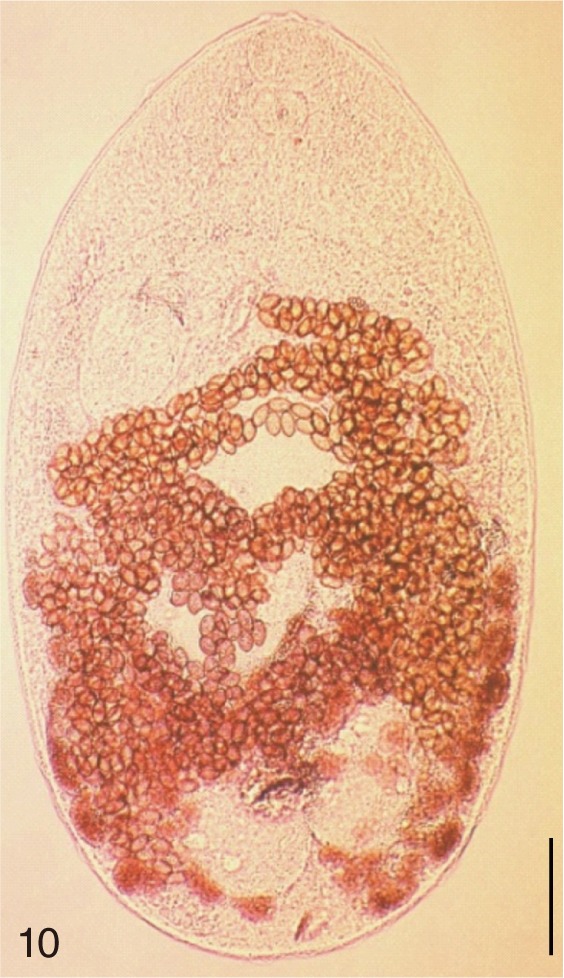
Fig. 11
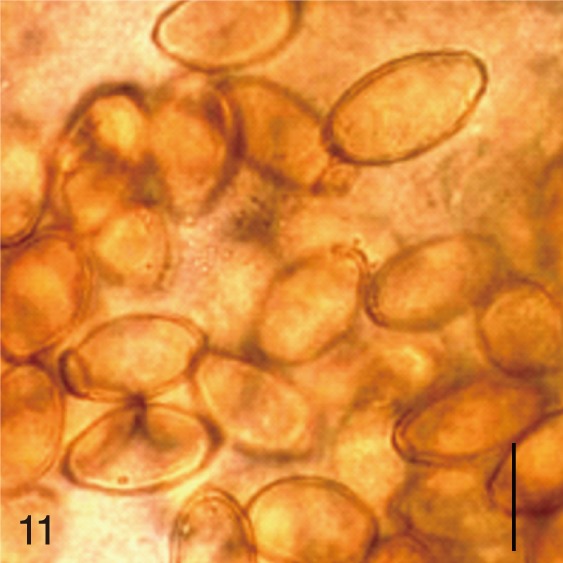
Metagonimus yokogawai. Uterine eggs. Bar=20 µm.
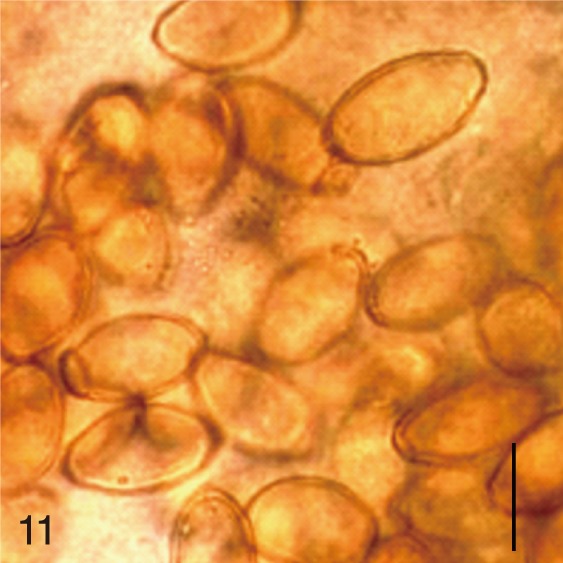
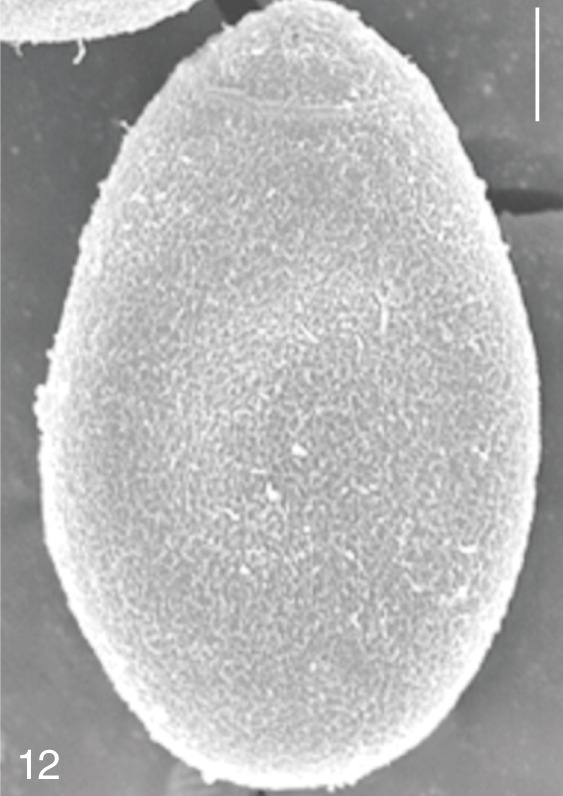
Fig. 12
Metagonimus yokogawai. SEM view of an egg showing relatively clean surface only with multiple minute ridges. Bar=4 µm.
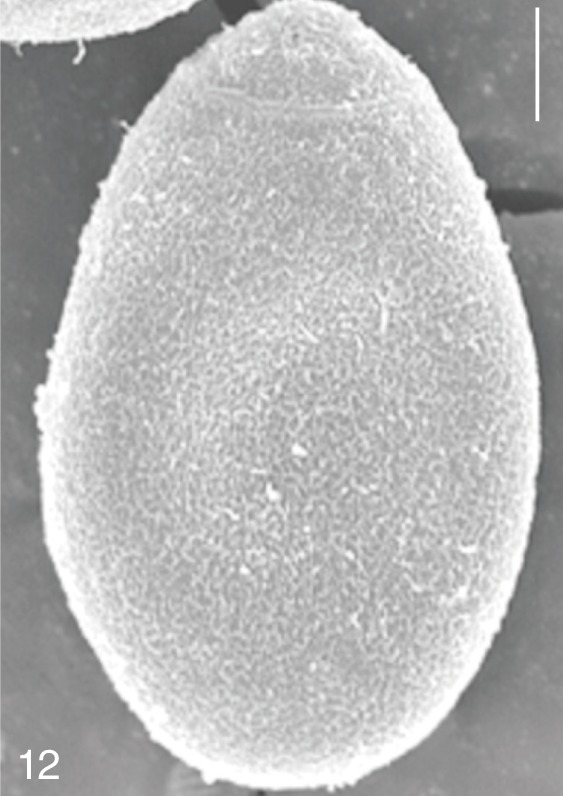
Fig. 13
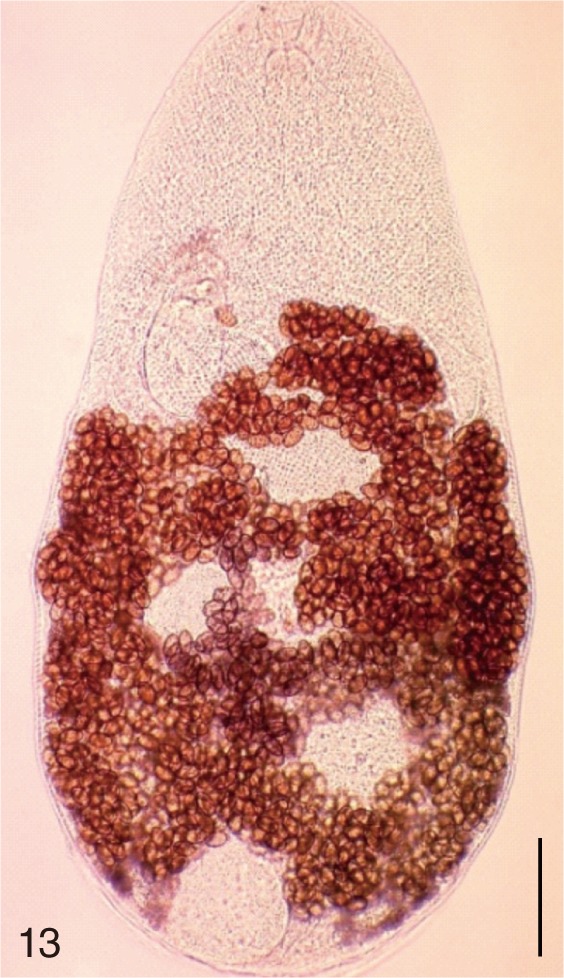
Metagonimus miyatai. An adult worm obtained from a naturally infected person in Umseong-gun, Korea. Bar=150 µm.
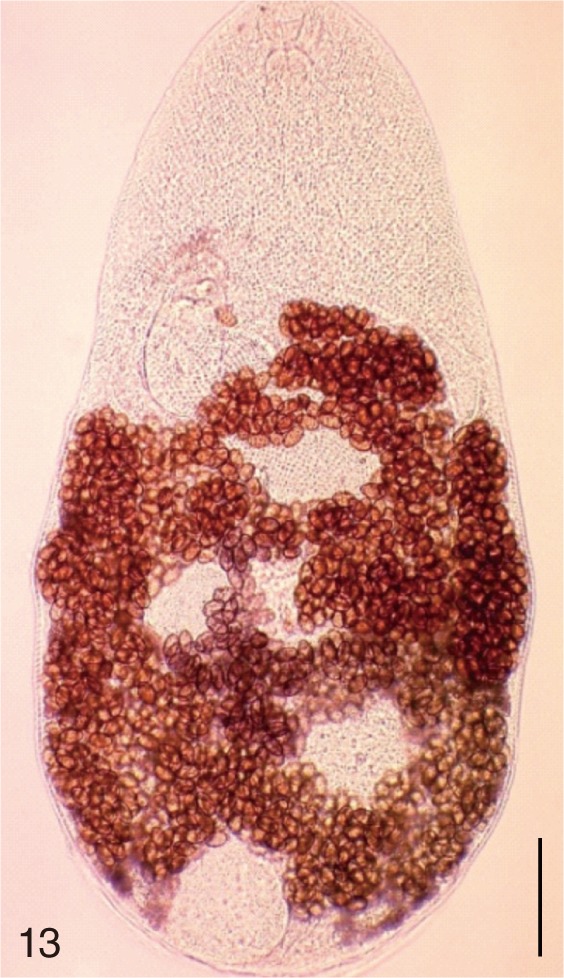
Fig. 14
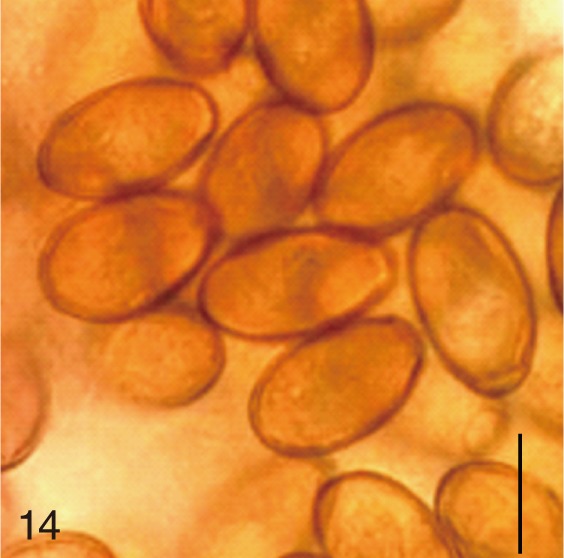
Metagonimus miyatai. Uterine eggs. Bar=20 µm.
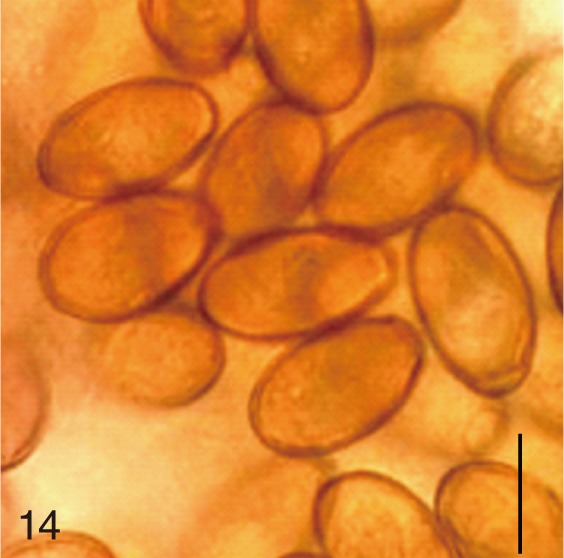
Fig. 15
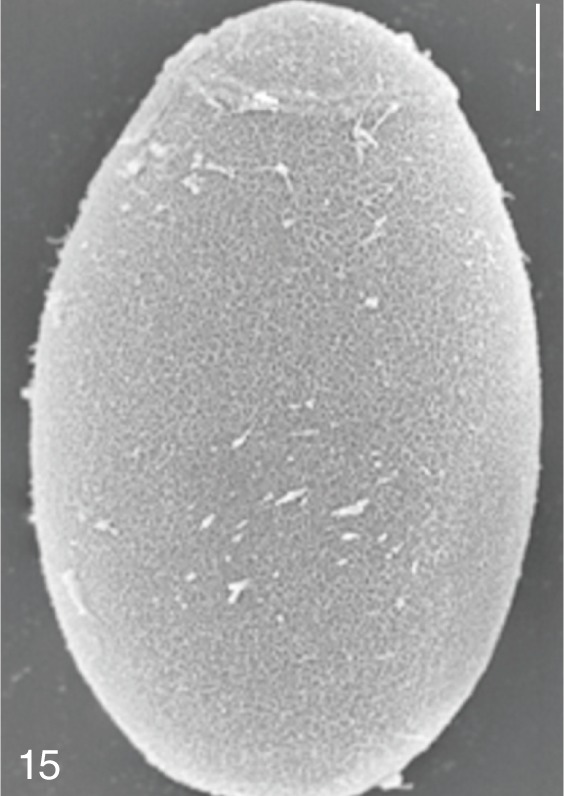
Metagonimus miyatai. SEM view of an egg showing relatively clean surface. Bar=4 µm.
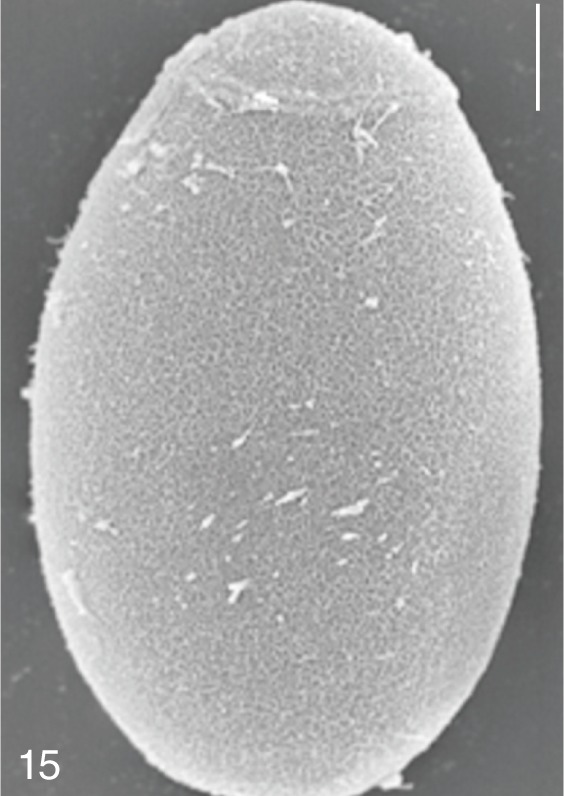
Fig. 16
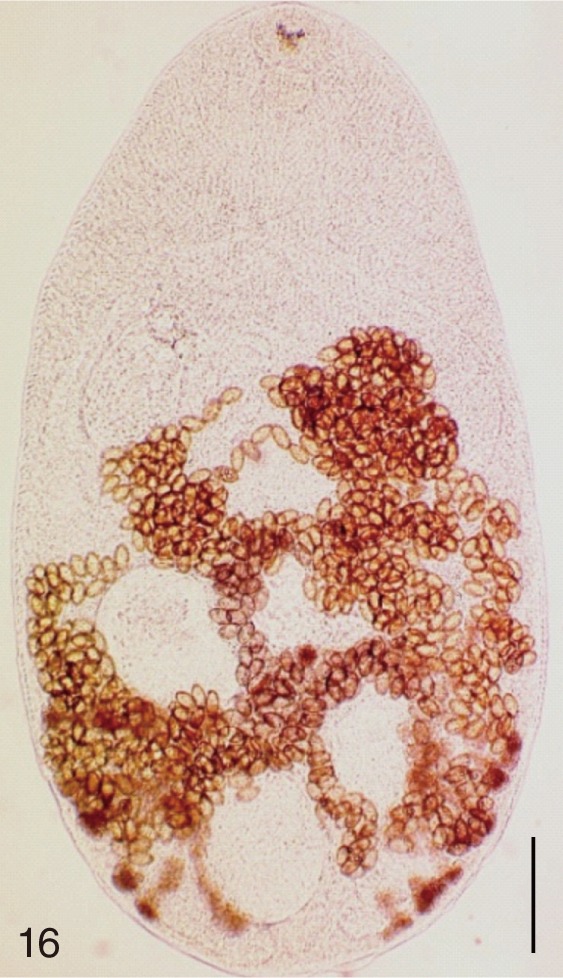
Metagonimus takahashii. An adult worm obtained from a naturally infected person in Umseong-gun, Korea. Bar=150 µm.
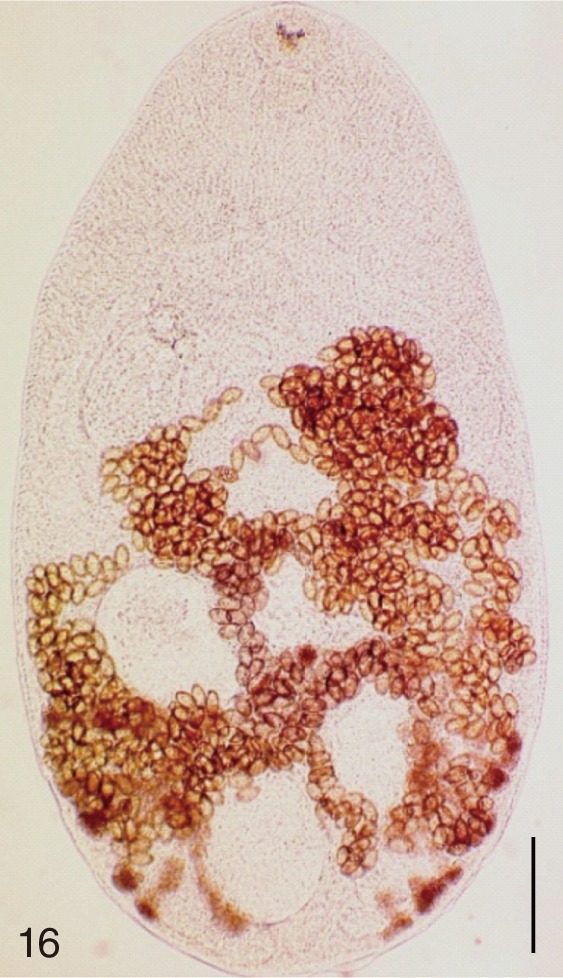
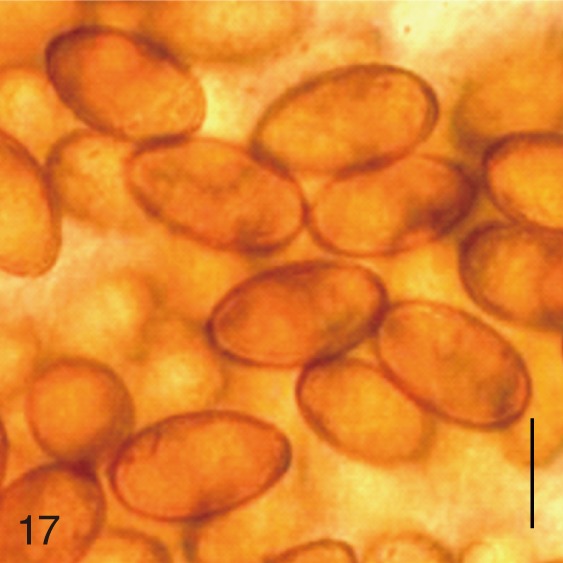
Fig. 17
Metagonimus takahashii. Uterine eggs. Bar=20 µm.
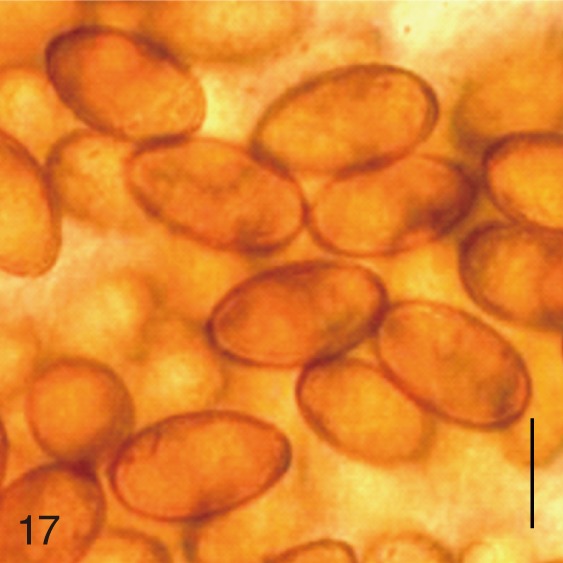
Fig. 18
Metagonimus takahashii. SEM view of an egg showing slightly rough surface. Bar=4 µm.

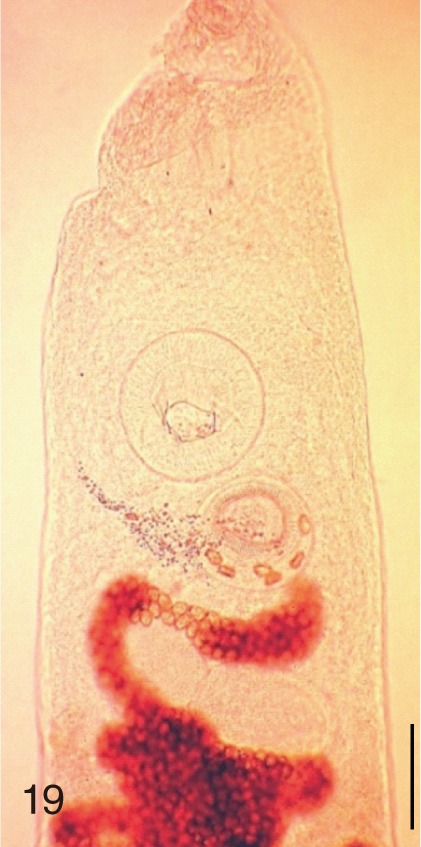
Fig. 19
Heterophyopsis continua. Anterior part of an adult worm obtained from a naturally infected person on Aphae-do, Korea. Bar=150 µm.
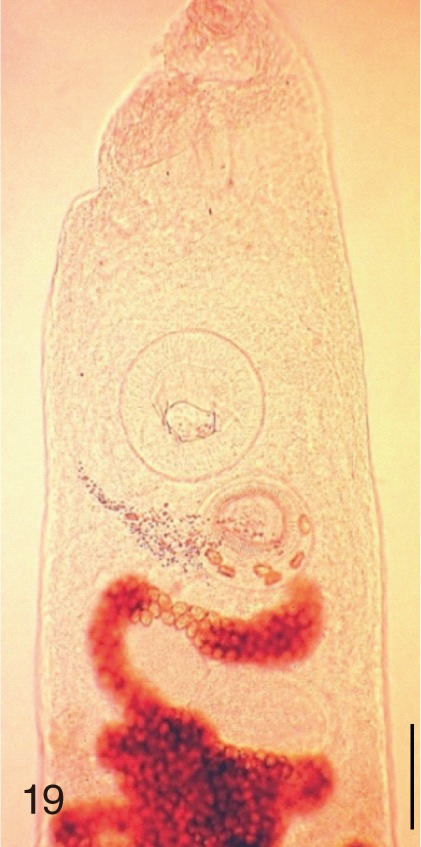
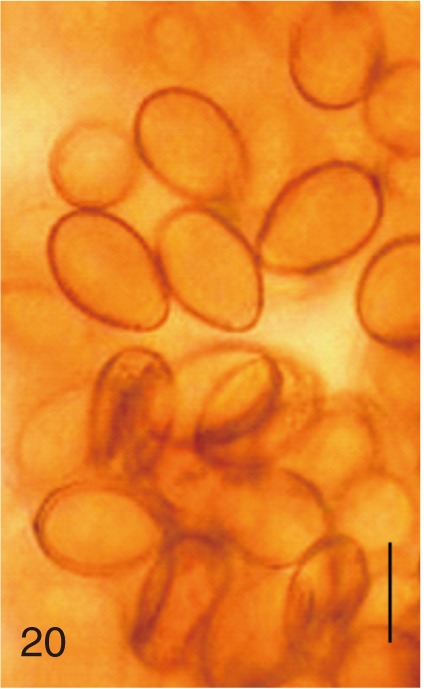
Fig. 20
Heterophyopsis continua. Uterine eggs. Bar=20 µm.
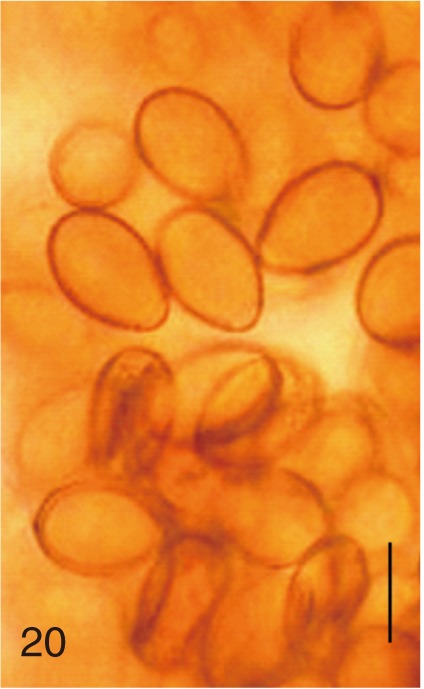
Fig. 21
Heterophyopsis continua. SEM view of an egg showing rough surface with a prominent operculum and shoulder rims. Bar=4 µm.
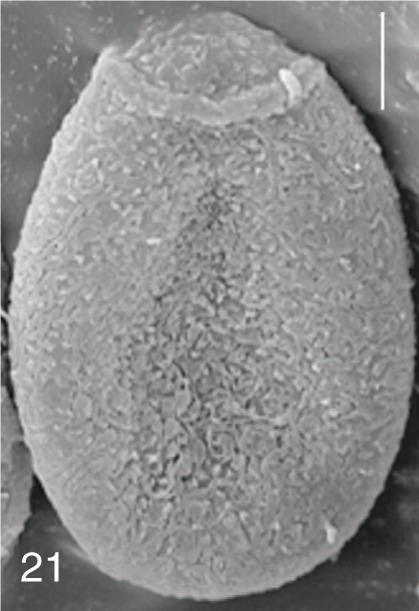
Fig. 22
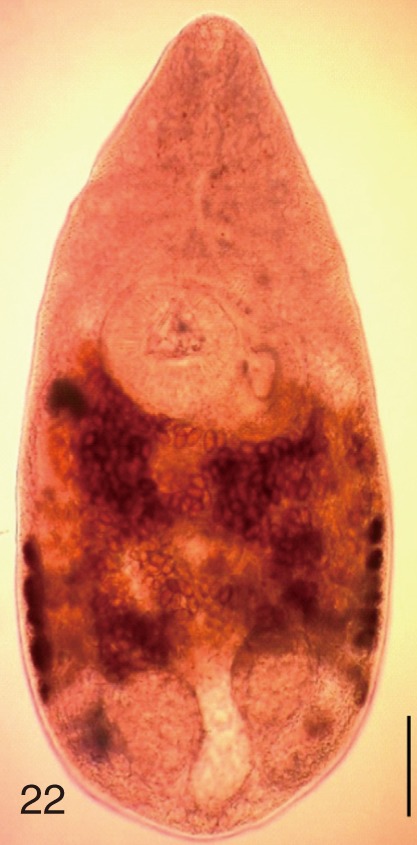
Heterophyes nocens. An adult worm obtained from a naturally infected person in Gangjin-gun, Korea. Bar=150 µm.
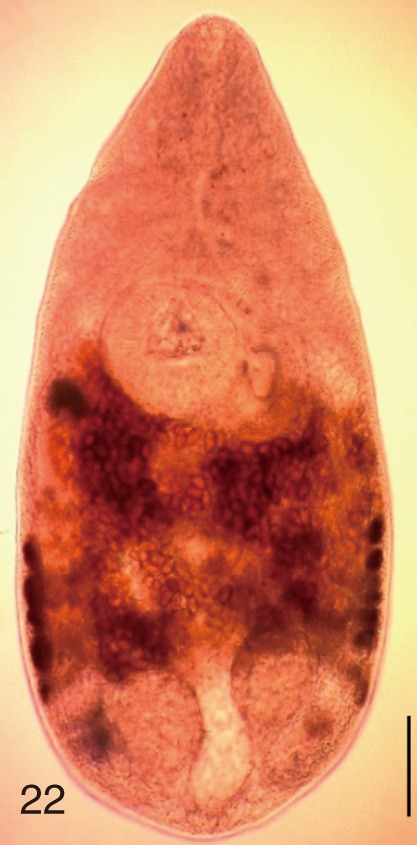
Fig. 23
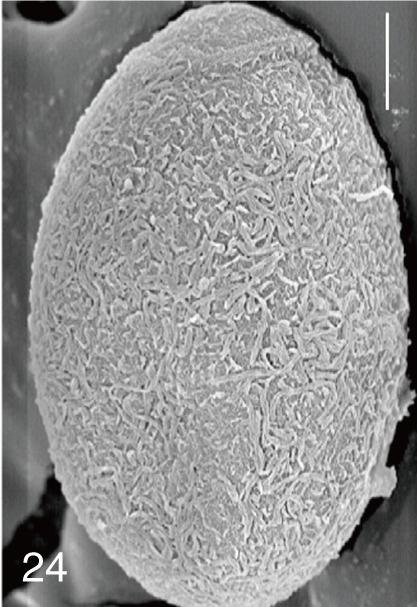
Heterophyes nocens. Uterine eggs. Bar=20 µm.

Fig. 24
Heterophyes nocens. SEM view of an egg showing rough surface with an inconspicuous operculum and shoulders. Bar=4 µm.

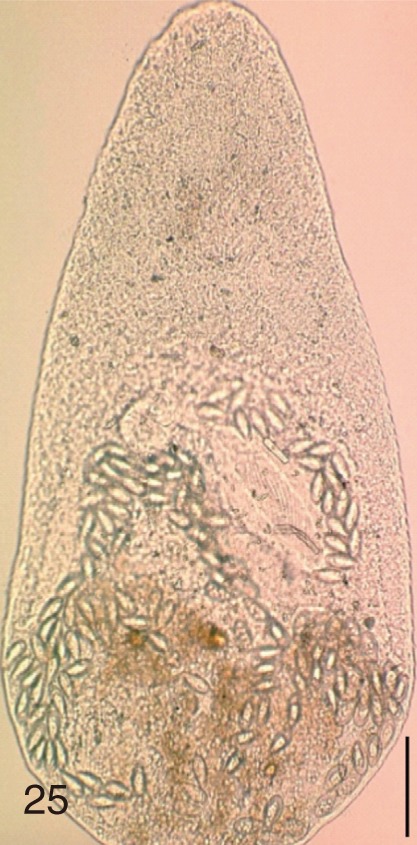
Fig. 25
Stellantchasmus falcatus. An adult worm obtained from a naturally infected person in Shinan-gun, Korea. Bar=80 µm.

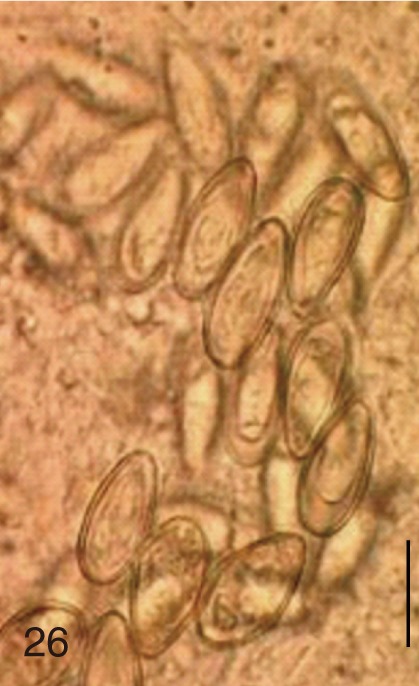
Fig. 26
Stellantchasmus falcatus. Uterine eggs. Bar=20 µm.
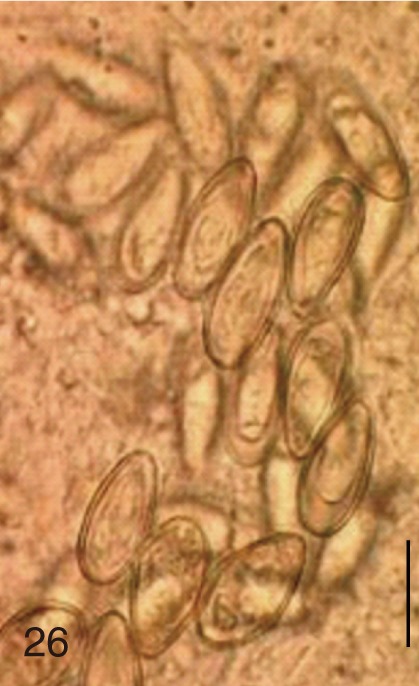
Fig. 27
Stellantchasmus falcatus. SEM view of an egg showing slightly rough surface and elongated shape. Bar=4 µm.

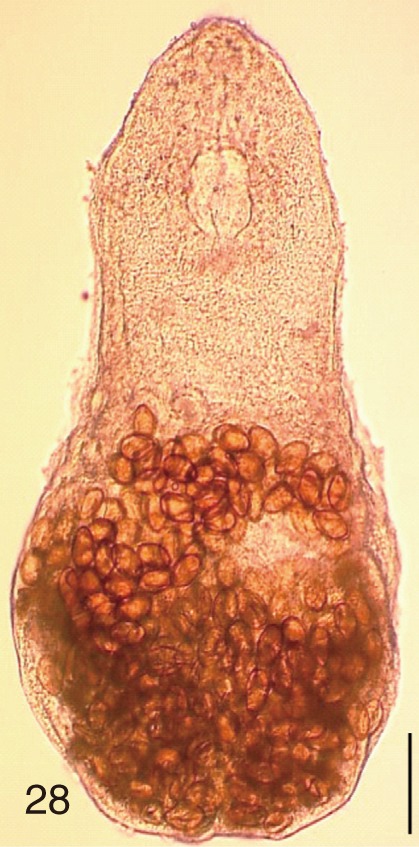
Fig. 28
Stictodora fuscata. An adult worm obtained from a naturally infected person in Shinan-gun, Korea. Bar=120 µm.
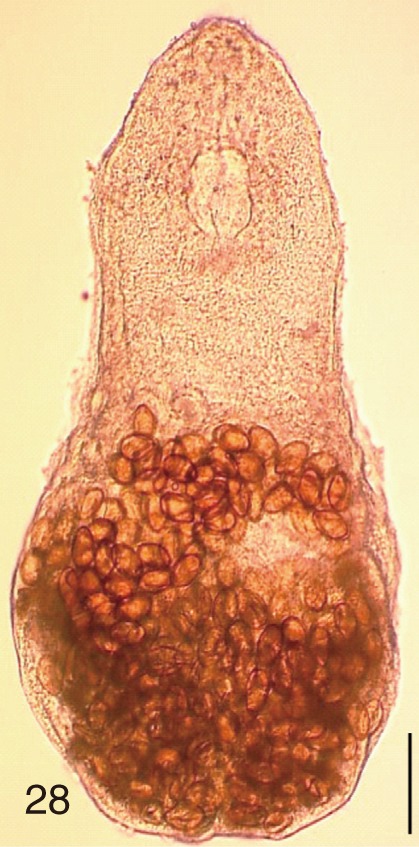
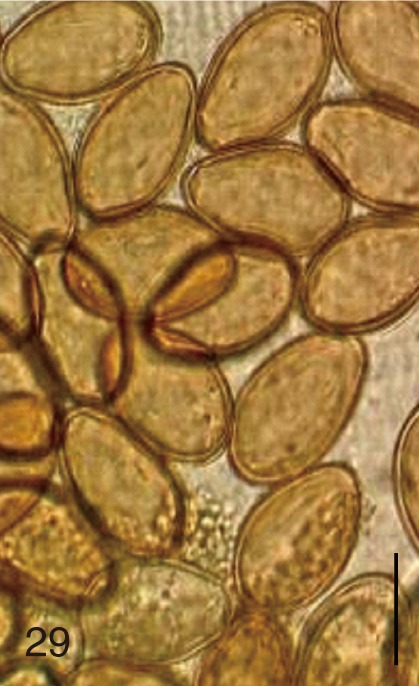
Fig. 29
Stictodora fuscata. Uterine eggs. Bar=20 µm.
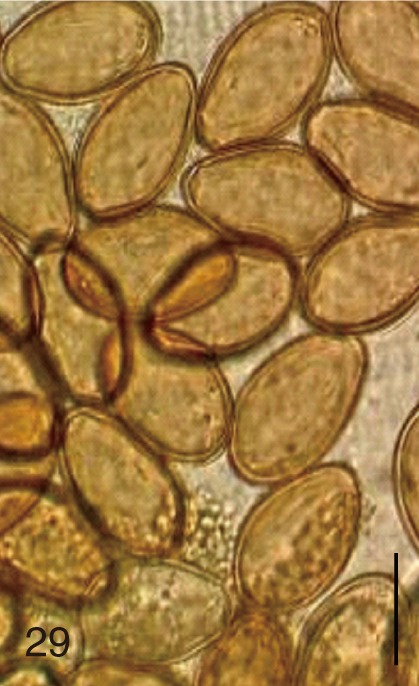
Fig. 30
Stictodora fuscata. SEM view of an egg showing clean surface and an inconspicuous operculum. Bar=4.5 µm.
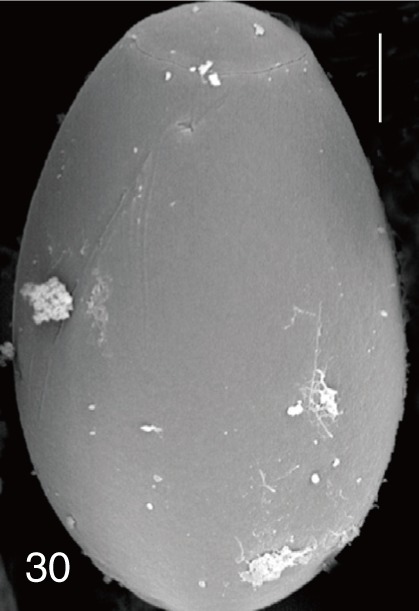
Fig. 31A diagram showing the range (mean±SD) of the egg length and FMI of minute intestinal flukes and Clonorchis sinensis occurring in Korea. Ps, Pygidiopsis summa; Gs, Gymnophalloides seoi; My, Metagonimus yokogawai; Mm, M. miyatai; Mt, M. takahashii; Hc, Heterophyopsis continua; Hn, Heterophyes nocens; Sfal, Stellantchasmus falcatus; Sfus, Stictodora fuscata; Cs, Clonorchis sinensis.

Table 1.Measurements of the eggs of minute intestinal flukes and C. sinensis that occur in the Republic of Korea
Table 1.
|
Fluke species |
Length (µm) |
Width (µm) |
FMIa
|
LWRb
|
|
Clonorchis sinensis
|
28.5 ± 1.3 |
15.7 ± 1.0 |
6,872 ± 593 |
1.8 ± 0.2 |
|
Pygidiopsis summa
|
20.9 ± 0.9 |
12.1 ± 1.0 |
3,046 ± 417 |
1.7 ± 0.2 |
|
Gymnophalloides seoi
|
22.9 ± 0.6 |
12.7 ± 0.5 |
3,681 ± 243 |
1.8 ± 0.1 |
|
Metagonimus yokogawai
|
28.4 ± 1.3 |
16.1 ± 1.4 |
7,380 ± 1,494 |
1.8 ± 0.1 |
|
Metagonimus miyatai
|
30.8 ± 0.9 |
17.8 ± 0.8 |
9,702 ± 1,042 |
1.7 ± 0.1 |
|
Metagonimus takahashii
|
32.6 ± 0.8 |
18.9 ± 1.3 |
11,646 ± 1,492 |
1.7 ± 0.1 |
|
Heterophyopsis continua
|
24.0 ± 1.0 |
15.5 ± 1.2 |
5,747 ± 757 |
1.6 ± 0.2 |
|
Heterophyes nocens
|
26.0 ± 1.6 |
14.1 ± 1.2 |
5,151 ± 975 |
1.9 ± 0.2 |
|
Stellantchasmus falcatus
|
26.3 ± 1.3 |
12.7 ± 0.6 |
4,223 ± 452 |
2.1 ± 0.1 |
|
Stictodora fuscata
|
33.6 ± 1.4 |
20.8 ± 0.7 |
14,608 ± 1,495 |
1.6 ± 0.1 |