Abstract
A 45-year-old man visited with suboccipital and posterior neck pain accompanied with blurred vision, dysarthria and paralytic feeling for 7 months. Magnetic resonance imaging revealed large eccentrically located cyst (6×4.8 cm) within posterior fossa which compressed right side cerebellar hemisphere and brainstem. The transparent cyst wall and fluid were removed by suboccipital craniectomy and the symptoms disappeared. Western blotting of the patient's cyst fluid reacted positively with the anti-cysticercus 150 kDa antibody and therefore the giant cyst was proved to be unusual giant neurocysticercus.
-
Key words: Neurocysticercosis, Taenia solium, giant cyst, Western blotting
Introduction
Neurocysticercosis (NCC) is a disease caused by infection of the central nervous system by
Taenia solium metacestode (TsM). Humans become infected by ingestion of
T. solium eggs through contaminated food and contact with carriers [
1]. Clinical manifestations of NCC vary depending on the location, number, and size of the cysts, the viability of the parasite, and the host’s inflammatory response including headache, seizure, focal neurologic deficits and hydrocephalus [
1-
3]. When the cysticerci are located within the basal cistern of cerebrospinal fluid (CSF), they may develop into a racemose form, characterized by a grape-like branching appearance. Histological studies have illustrated that the cyst bladder wall can proliferate into several centimeters in size after the scolex has degenerated [
4]. Although the mechanism underlying this transformation has not been clearly elucidated, it is generally accepted that continuous influx of CSF inside the cysts leads to degeneration of the scolex and progressive proliferation of the bladder wall [
4]. Some cysticerci within the Sylvian fissure or the basal cistern of CSF may enlarge up to 5 cm or more, forming giant cyst similar to racemose type [
4]. Recently, several clinical cases of giant NCC have been reported based on radiological findings and surgical treatments [
5-
8], however, cases with immunodiagnostic confirmation are rarely documented. Moreover, the protein composition and properties of the cyst fluid (CF) collected during surgical procedures have not been well characterized.
In this study, we attempted to detect and diagnose NCC using the CF obtained from surgically collected patient cyst by Western blot analysis.
Case Report
In November 2006, a 45-year-old man was presented to Jeju National University Hospital with suboccipital and posterior neck pain. The patient had complained episodic blurred vision, dysarthria and paralytic feeling for 7 months. Blurred vision had lasted only a few seconds in binocular or monocular fashion. On the other hand, dysarthria and paralytic feeling had lasted longer, sometimes reaching an hour. Symptoms had appeared irregularly, often several times a day. During examination, the patient did not complain any symptoms. Mild hypoesthesia (about 20% loss) was detected at scattered area on the patient's right side face and body. No subcutaneous nodules were detected. No risk factors for stroke was identified except for smoking (20 pack-year). The patient had never eaten raw pork meat neither had family history of tapeworm infection or cysticercosis.
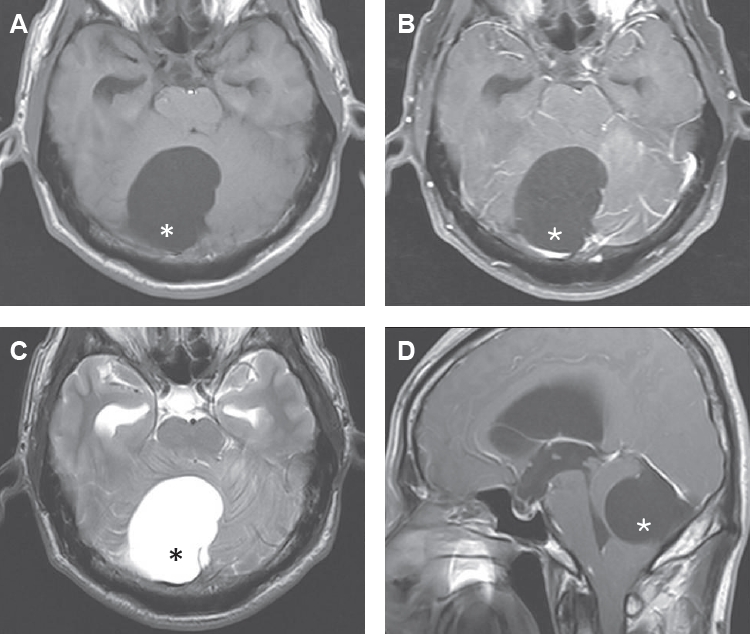
Magnetic resonance imaging (MRI) showed large eccentrically located cyst (6×4.8 cm) within posterior fossa which massively compressed right cerebellar hemisphere and brainstem (
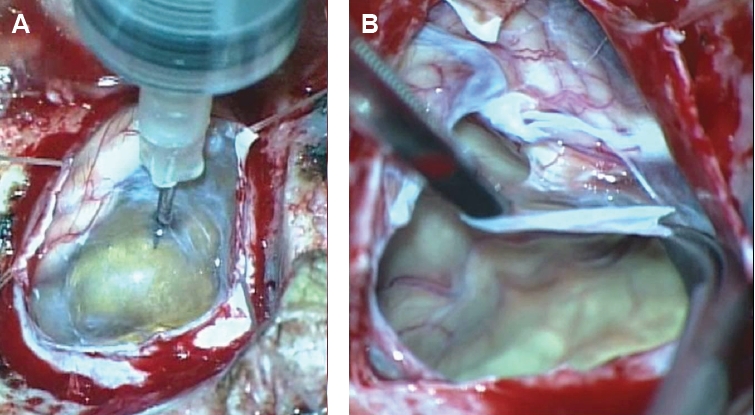
Fig. 1). The cyst was sharply delineated from the cerebellum and compressed the fourth ventricle, resulting in ventricular dilatation (i.e., obstructive hydrocephalus). The cerebellar tonsil was herniated through foramen magnum. The CF exhibited signal intensity identical to that of CSF on MRI, and the cyst wall showed no enhancement on gadolinium-enhanced images. Accordingly, the MRI findings favored a diagnosis of an arachnoid cyst in the posterior fossa. However, the location of the cyst was quite different from that of a typical arachnoid cyst, which is usually located in midline. Because the cyst caused symptomatic hydrocephalus, the patient underwent surgery. Under general endotracheal anesthesia, midline suboccipital craniectomy over the cyst was performed. After incising the dura, thin and transparent cyst wall was exposed freely. Arachnoid membrane was integrated with cyst wall. The authors aspirated cystic fluid using a 23-gauge needle (
Fig. 2A). The CF was clear and colorless, therefore macroscopic findings could not distinguish from CSF. Freely exposed cyst wall (posterior part of the cyst) was widely removed (
Fig. 2B) and pathologically examined. The authors were unable to dissect the cyst wall from the cerebellum (anterior and lateral portions of the cyst) because the cyst wall, arachnoid membrane, and pia mater were fused in this region. There was no visible evidence of a scolex within the cyst, and the patient’s symptoms completely disappeared following surgical treatment. No anthelminthic drugs were administered after surgery. Thereafter, he did not experience blurred vision or any neurological deficits.
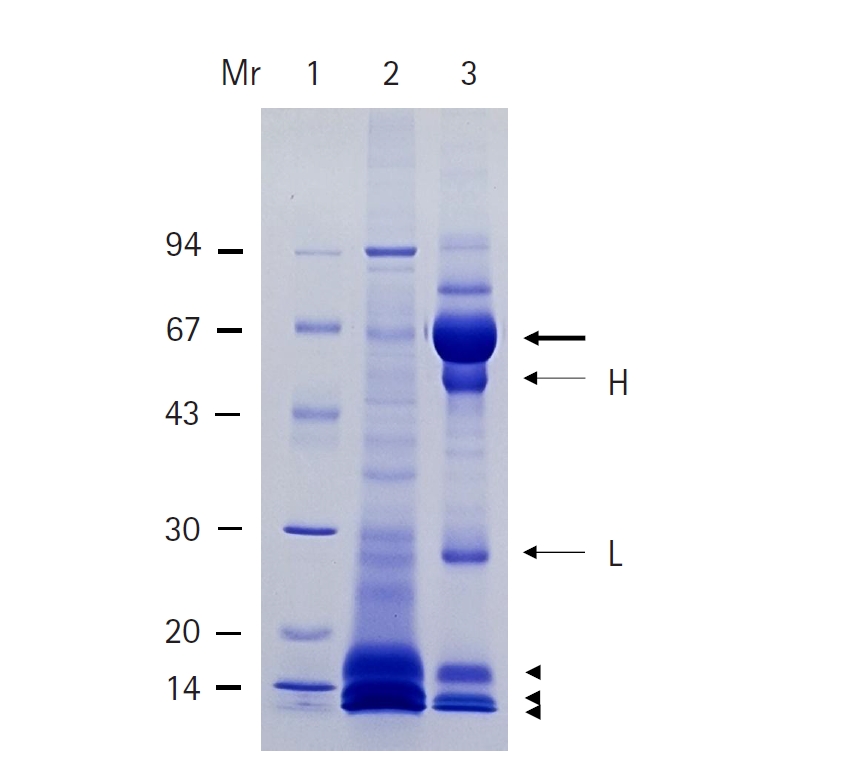
Electrophoretic analysis of CF from the patient and pig revealed distinct protein profiles. CF collected from pigs exhibited protein bands distributed across a wide range of molecular weights, with particularly abundant low molecular weight fractions at 7, 10, and 15 kDa. In contrast, patient-derived CF showed a predominant band at 66 kDa, along with prominent bands at 55 and 28 kDa; appreciable amounts of the 7, 10, and 15 kDa fractions were also detected (
Fig. 3).
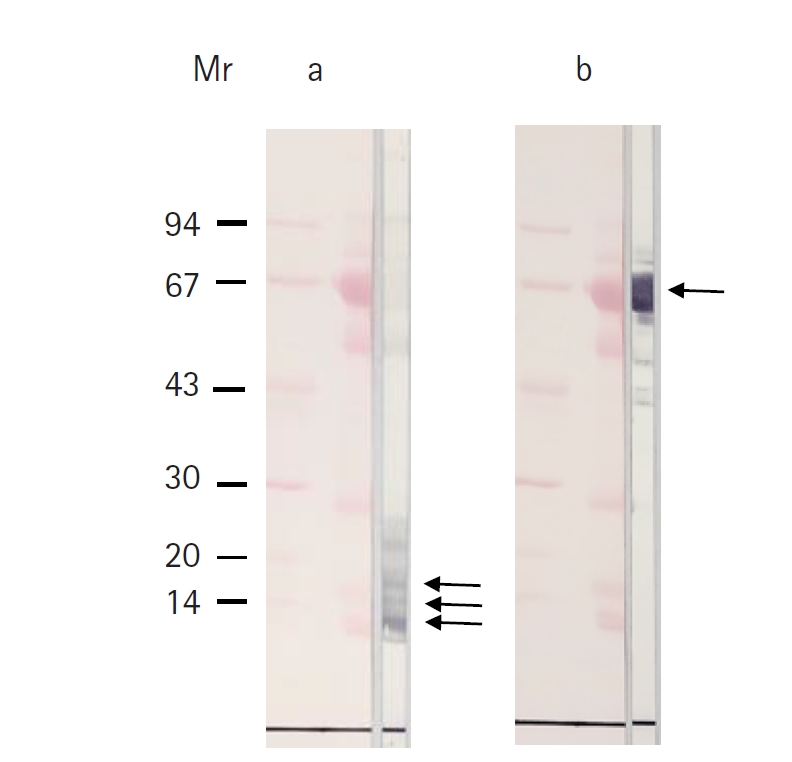
Western blot analysis demonstrated that the predominant 66 kDa band in patient-derived CF reacted with a monoclonal antibody against human albumin, confirming its identity as albumin. In addition, the 7, 10, and 15 kDa bands specifically reacted with an antibody against the 150 kDa antigen unique to TsM (
Fig. 4).
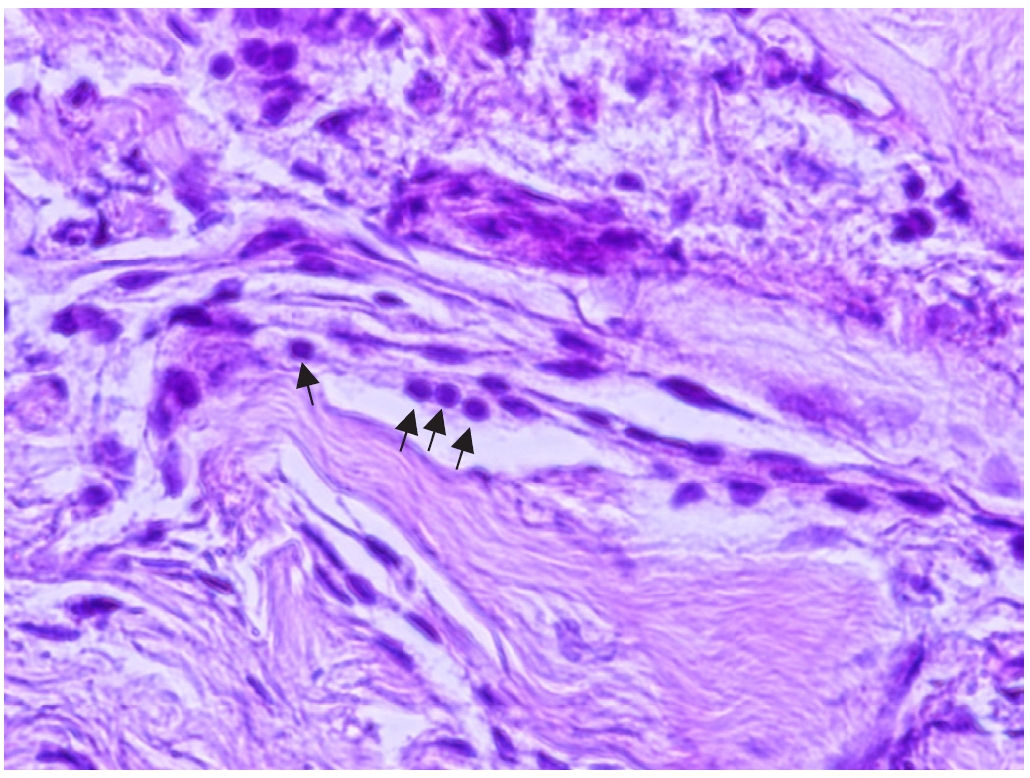
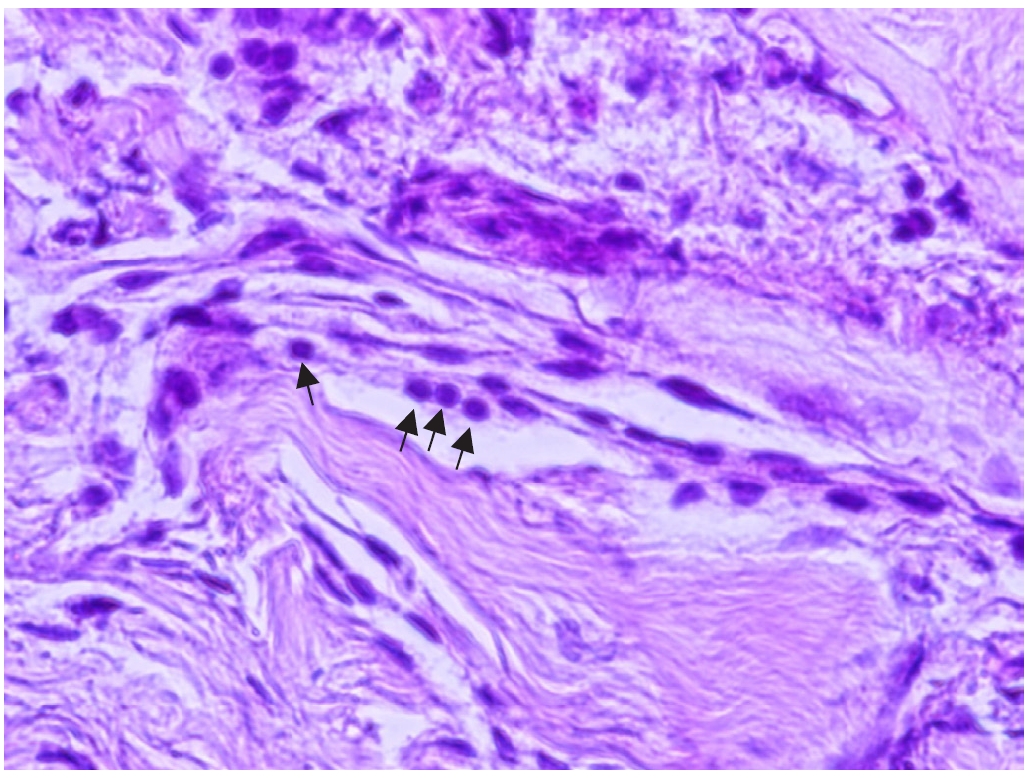
Histopathological examination revealed irregular, multilayered fibrous structures accompanied by numerous calcareous corpuscles (
Fig. 5). This study was approved by the Institutional Review Board of Jeju National University Hospital (JEJUNUH 2026-01-037) and informed consent was waived.
Discussion
In general, the diagnosis of NCC is based on radiological findings, immunological tests, epidemiological evidence from endemic areas, and the patient’s clinical history [
3]. In the present study, MRI findings demonstrated that the cyst exhibited signal intensity identical to that of CSF even after gadolinium contrast administration, consistent with previous reports that giant cysts usually have the same signal properties and do not exhibit enhancement after gadolinium administration [
4]. This imaging characteristic is likely attributable to continuous influx of CSF into the cyst. Furthermore, the cyst appeared as a single large cyst without internal septation or conglomeration, and no scolex was identified on macroscopic examination, distinguishing it from the racemose form of NCC.
Analysis of SDS-PAGE showed that the overall patterns of the patient CF were somewhat different from that of TsM (
Fig. 3). CF of TsM consisted of bands ranging from 94 to 7 kDa bands and showed major protein bands of 15, 10, and 7 kDa. However, CF derived from the patient revealed major protein bands of 66, 55, 28, 15, 10, and 7 kDa. Among these bands, a 66 kDa band showed a highly enriched amount and may correspond to human serum albumin [
9]. The 55 and 28 kDa protein bands were also detected and seemed likely to be heavy and light chains of human IgG, respectively [
9]. The 15, 10, and 7 kDa protein bands observed in the patient CF showed a pattern similar to those of TsM CF but were present at much lower levels (
Fig. 3).
Western blotting studies of the patient CF using a specific antibody to the 150 kDa protein of TsM CF revealed positive reactivity with the 15, 10, and 7 kDa protein bands in the patient’s CF (
Fig. 4a). The 150 kDa protein of TsM is known to be a specific diagnostic antigen for NCC and a lipid-binding protein that plays an important role for survival and homeostasis of TsM [
10]. In this regard, the 150 kDa protein was clearly detected in the patient-derived CF, indicating cysticercosis infection. Another major protein of 66 kDa band of the patient CF was confirmed to be human serum albumin using a monoclonal antibody against human serum albumin (
Fig. 4b). This is consistent with the previous study of Kong et al. [
9], which described that serum albumin and IgG were also observed on SDS-PAGE protein staining in CF of NCC patients, including racemose cysticercus. It is likely that human serum albumin and immunoglobulin G were contaminated during surgical collection, such as aspiration with a syringe. The authors were able to collect CF from the patient and use it for diagnostic purposes, however, serological diagnostic methods such as ELISA are generally useful in routine clinical practice.
Microscopic examination showed irregular, multilayered fibrous structures accompanied by numerous calcareous corpuscles, which were histologically distinct from the racemose form. Calcareous corpuscles are well recognized as important histological markers of cestode tissue [
11].
In the present study, the authors identified the patient’s cyst as giant NCC through radiological, histochemical, and serological analyses particularly using a specific anti-150 kDa antibody from TsM.
Notes
-
Author contributions
Conceptualization: Lee CS, Chung YB. Funding acquisition: Chung YB. Investigation:Lee CS, Lee YA, Chung YB. Methodology: Lee CS, Lee YA, Koh HM, Chung YB. Supervision:Chung YB. Visualization: Lee CS, Lee YA, Koh HM, Chung YB. Writing – original draft: Lee CS, Chung YB. Writing – review & editing: Lee CS, Lee YA, Chung YB.
-
Conflict of interest
The authors have no conflicts of interest to declare.
-
Funding
This work was supported by the grant 2025 education, research and student guidance grant funded by Jeju National University.
Fig. 1.Magnetic resonance images show hydrocephalus and a large cyst in the posterior fossa with signal intensity identical to cerebrospinal fluid and no contrast enhancement after Gd-DTPA injection. Asterisks show a large cyst in the posterior fossa. T1 axial image (A), T1 gadolinium enhance image (B), T2 axial image (C), and T1 sagittal image (D).
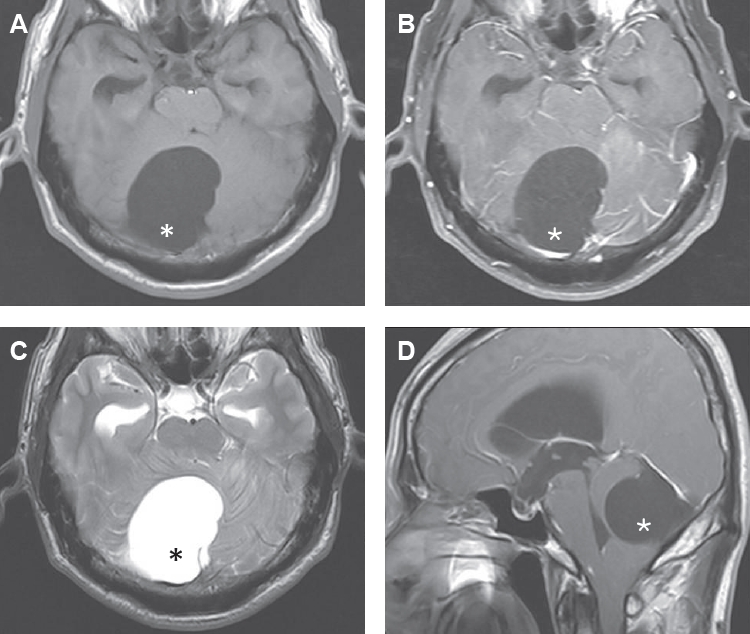
Fig. 2.The cyst fluid of the transparent membranous cyst was aspirated with a 23-gauge needle (A). Thereafter, the cyst wall was widely removed (B).
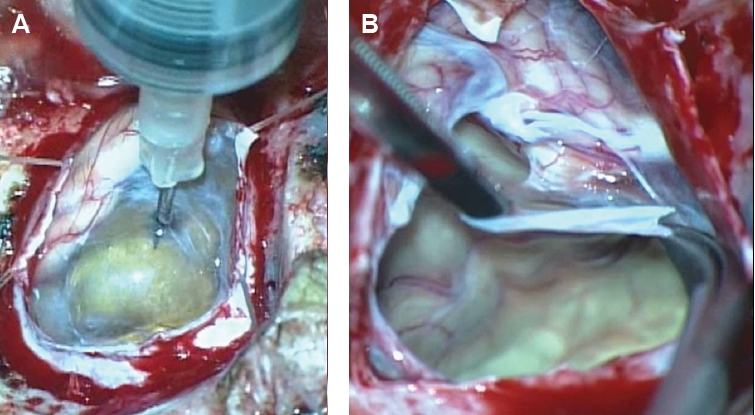
Fig. 3.SDS-PAGE finding of cyst fluid (CF) from Taenia solium metacestode and the patient. Lane 1, standard marker proteins; lane 2, CF of T. solium metacestode; lane 3, CF of the patient. The bold arrow indicates the serum albumin from the patient’s CF. H and L indicate the heavy and light chains of IgG, respectively. Arrowheads indicate 15, 10, and 7 kDa protein bands. SDS-PAGE was performed using 7.5%–15% gradient separating gel with 35 mA constant current. After electrophoresis, the gel was stained with Coomassie brilliant blue R250. Standard molecular weight marker proteins indicate band at 94, 67, 43, 30, 20, and 14 kDa. Mr, molecular weight (kDa).
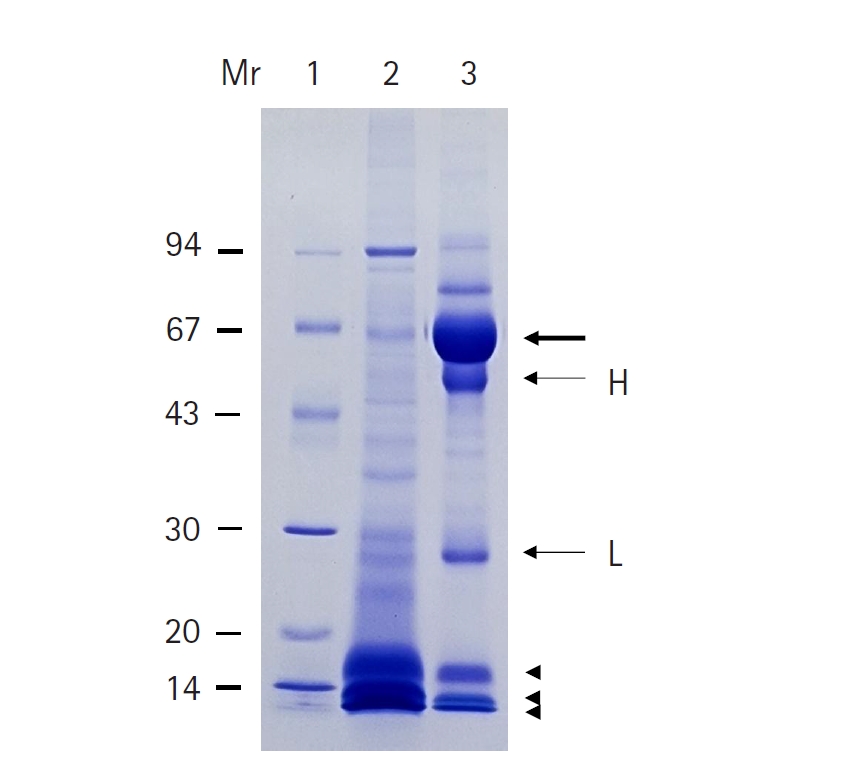
Fig. 4.Immunoblot analysis of the cyst fluid (CF) from the patient. (a) The CF was reacted with anti-150 kDa antibody of
Taenia solium metacestode. Positive reactions were observed at 15, 10, and 7 kDa protein bands (arrows). (b) The CF was probed with a monoclonal antibody against human serum albumin. Albumin (66 kDa) of the CF reacted strongly with the antibody (arrow). The transferred standard marker proteins and proteins of the CF were visualized with Ponceau S staining. The crude extracts of the patient’s CF were separated using 7.5%–15% gradient SDS-PAGE gel. The gel was transferred onto polyvinylidene fluoride (Millipore) membranes for 1 h with 100 V. The transferred blots were then incubated with a specific anti-150 kDa antibody [
9] and a mouse monoclonal anti-human serum albumin antibody (Thermo Fisher Scientific) for 2 h at a dilution of 1:100. The blots were washed with phosphate buffered saline/Tween 20 3 times. The blots were further incubated with secondary horseradish peroxidase–conjugated antibodies (Cappel) for 2 h at a dilution of 1:1,000 and developed with 4-chloro-1-naphthol.
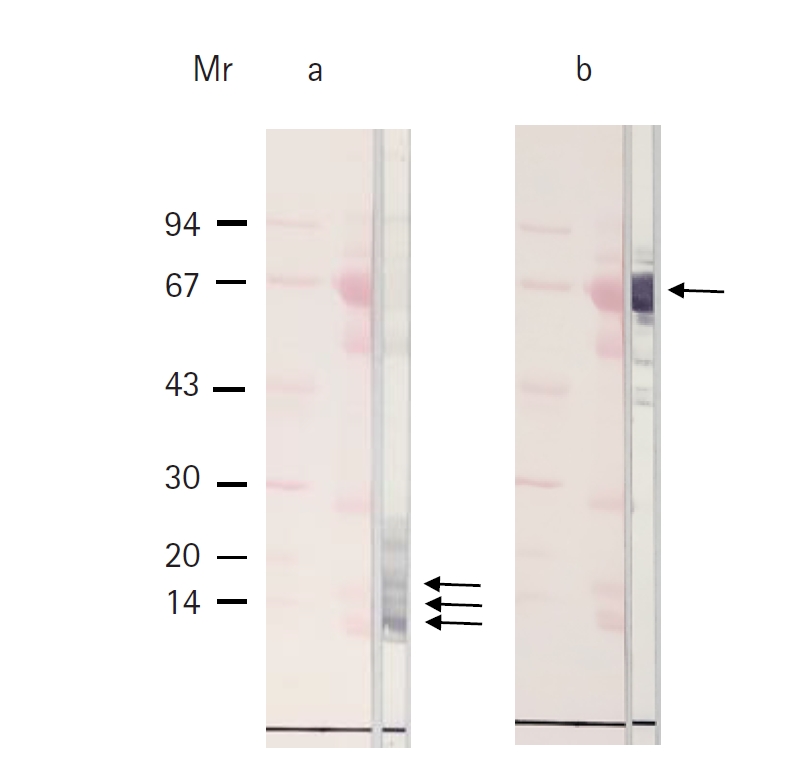
Fig. 5.Microscopic examination revealed an irregular, multifold, fibrous-like layer, and numerous calcareous corpuscles were observed (×200). Hematoxylin-eosin staining was used and arrows indicate calcareous corpuscles.
References
- 1. Del Brutto OH. Meningeal cysticercosis. In Singh G, Prabhakar S, editors. Taenia solium cysticercosis: from bascic to clinical science. CABI Publishing; 2002. p. 177-97.
- 2. Garcia HH, Nash TE, Del Brutto OH. Clinical symptoms, diagnosis, and treatment of neurocysticercosis. Lancet Neurol 2014;13:1202-15. http://doi.org/10.1016/S1474-4422(14)70094-8
- 3. Del Brutto OH. Human neurocysticercosis: an overview. Pathogens 2022;11:1212. http://doi.org/10.3390/pathogens11101212
- 4. Del Brutto OH, Sotelo J, Román GC. Neurocysticercosis: a clinical handbook. Swets & Zeitlinger; 1988.
- 5. Kim JH, Suh SI, Kim JH, Kwon TH, Chung HS. Giant neurocysticercosis cyst in the cerebellar hemisphere. Neurol Med Chir (Tokyo) 2006;46:412-4. http://doi.org/10.2176/nmc.46.412
- 6. Ramesh VG, Parthiban A. Giant parenchymal cysticercosis with unusual imaging features. J Clin Neurosci 2008;15:1404-6. http://doi.org/10.1016/j.jocn.2007.09.026
- 7. Proaño JV, Madrazo I, Avelar F, et al. Medical treatment for neurocysticercosis characterized by giant subarachnoid cysts. N Engl J Med 2001;345:879-85. http://doi.org/10.1056/NEJMoa010212
- 8. Zumaeta J, Contreras C, Tapia P, et al. Giant neurocysticercosis: a rare medical condition. Cureus 2024;16:e71090. http://doi.org/10.7759/cureus.71090
- 9. Kong Y, Kang SY, Cho SY. Component proteins in cystic fluid of Taenia solium metacestodes collected surgically from neurocysticercosis patients. Korean J Parasitol 1990;28:101-8. https://doi.org/10.3347/kjp.1990.28.2.101
- 10. Lee EG, Kim SH, Bae YA, et al. A hydrophobic ligand-binding protein of the Taenia solium metacestode mediates uptake of the host lipid: implication for the maintenance of parasitic cellular homeostasis. Proteomics 2007;7:4016-30. http://doi.org/10.1002/pmic.200700332
- 11. Gutierrez Y. Diagnostic pathology of parasitic infections with clinical correlations. 2nd ed. Oxford University Press; 2000.
Citations
Citations to this article as recorded by