Abstract
Strongyloidiasis is a neglected infectious disease with diverse clinical manifestations that may lead to severe complications, including death in immunocompromised individuals. Uncomplicated infections can be diagnosed early using serological testing. Regarding treatment, the World Health Organization recommends ivermectin, thiabendazole, and albendazole as effective therapies against Strongyloides stercoralis. The objective of this study was to evaluate the therapeutic effectiveness of ivermectin 200 µg/kg/day for 2 consecutive days in patients infected with S. stercoralis, with the aim of optimizing treatment efficacy while shortening the duration of therapy compared with longer regimens. This before-and-after study assessed the therapeutic effectiveness of ivermectin 200 µg/kg/day for 2 consecutive days in patients with S. stercoralis infection. From January to October 2023, 3,814 patients with suspected S. stercoralis infection—presenting with symptoms such as urticaria, abdominal pain, and loose stools with seropositivity on an enzyme-linked immunosorbent assay for IgG antibodies against S. stercoralis—were screened, and 42 patients with uncomplicated S. stercoralis mono-infection were recruited. Symptoms, IgG optical density antibody titers against S. stercoralis, and eosinophils were assessed before and after treatment. Therapeutic effectiveness was evaluated based on cure rate and disease reduction after intervention. After 3 months of treatment, the overall effectiveness of ivermectin 200 µg/kg/day for 2 consecutive days reached 95.2%, of which 50% were completely cured, 9.5% had symptom resolution without improvement in paraclinical indicators, 21.4% experienced clear symptom reduction, and 14.3% had symptom recurrence. The regimen of ivermectin 200 µg/kg/day for 2 consecutive days demonstrated high effectiveness in the treatment of strongyloidiasis.
-
Key words: Strongyloides stercoralis, strongyloidiasis, ivermectin
Introduction
Strongyloides spp. is a genus of nematodes that parasitize the digestive tract of vertebrates, of which 2 species—
S. stercoralis and
S. fuelleborni—are capable of causing disease in humans. Infection occurs through contact with soil contaminated with feces containing infective larvae that penetrate the skin. The global prevalence of
Strongyloides infection has not been precisely determined; however, experts estimated that in 2017 the global infection rate was 8.1%, corresponding to approximately 613.9 million people worldwide, with higher prevalence in Southeast Asia, Africa, and the Western Pacific [
1]. In Vietnam, reported seropositivity rates for
S. stercoralis at parasitology medical facilities—including Hanoi Medical University Hospital, the Institute of Malariology, Parasitology and Entomology Quy Nhon, and the University Medical Center Ho Chi Minh City—were 46.3%, 4.76%, and 7.9%, respectively, indicating a substantial infection burden [
2-
4].
Strongyloidiasis is frequently underdiagnosed or diagnosed late because many infections are asymptomatic and stool-based larval detection methods have low sensitivity. Delayed diagnosis may result in severe complications, including hyperinfection syndrome and disseminated strongyloidiasis, which are associated with high mortality. With advances in immunological diagnostics,
S. stercoralis infection can now be detected earlier in uncomplicated cases, often presenting with the classic triad of diarrhea, abdominal pain, and itchy urticaria. For treatment, the World Health Organization recommends ivermectin, thiabendazole, and albendazole as effective therapies against
S. stercoralis [
5]. Ivermectin is a semi-synthetic anthelmintic derived from
Streptomyces avermitilis. It enhances γ-aminobutyric acid–mediated neurotransmission, increases chloride influx into parasite cells, and causes hyperpolarization and paralysis of worm musculature. Ivermectin is effective for strongyloidiasis,
Onchocerca volvulus infection, other helminthic diseases, and ectoparasitic infestations such as scabies, lice, and
Demodex spp. [
6]. Previous studies evaluating ivermectin regimens have reported variable effectiveness. A regimen repeated every 2 weeks at a dose of 200 µg/kg/day was not more effective than a single treatment [
7]. Multiple-dose regimens administered on days 1, 2, 15, and 16 demonstrated comparable therapeutic efficacy and adverse-effect profiles to a single 200 µg/kg dose [
8]. At the time of this study, the efficacy and safety of ivermectin 200 µg/kg/day administered for 2 consecutive days had not been reported. Therefore, to optimize efficacy while reducing treatment duration and cost compared with multiple-dose regimens, this study aimed to evaluate the therapeutic effectiveness of ivermectin 200 µg/kg/day for 2 consecutive days in patients with uncomplicated
S. stercoralis infection.
Methods
The study was approved by the Protocol Approval Board of the Department of Microbiology–Parasitology, Faculty of Medicine, University of Medicine and Pharmacy at Ho Chi Minh City, and by the Biomedical Ethics Board of the Institute of Malariology, Parasitology and Entomology Quy Nhon, Vietnam (approval No. 1141/VSR-HDDD dated December 28, 2022), prior to implementation. Diagnosis, treatment, and monitoring of S. stercoralis–infected cases at IMPE Quy Nhon followed the 2022 guidelines of the Vietnam Ministry of Health.
This before-and-after study design evaluated the cure rate and disease reduction after intervention with ivermectin 200 µg/kg/day administered for 2 consecutive days.
Patients diagnosed with isolated S. stercoralis infection at IMPE Quy Nhon from January to October 2023 were screened from 3,814 individuals diagnosed with S. stercoralis infection for recruitment. Eligible participants had a positive ELISA IgG test against S. stercoralis with optical density (OD) ≥1 and were negative for other helminths, including Toxocara canis, Gnathostoma spinigerum, Fasciola spp., and Cysticercus cellulosae. Participants were required to have at least 1 disease-related symptom (e.g., itching, urticaria, diarrhea, abdominal pain, digestive disorders), no use of specific anti-helminthic treatment within the previous 6 months, and to provide informed consent. Exclusion criteria included contraindications to ivermectin (age <6 years or weight <15 kg), pregnancy or lactation, and underlying conditions potentially affecting treatment effectiveness, including cancer, asthma, diabetes, collagen vascular disease, systemic disease, pulmonary tuberculosis, renal failure, blood–brain barrier disorders, meningitis, or other neuropsychiatric conditions.
Sample size for the before-and-after design was calculated using the following formula:
n=z1-α/22p(1-p)d2. Based on previous findings at the same site reporting a cure and disease-reduction rate of 84.6% with single-dose ivermectin [
9], the expected rate for the 2-day regimen was set at
P=0.9. With α=5% and d=0.1, the calculated sample size was 35. To account for potential loss to follow-up and ineligible cases, the sample size was increased by 20%, yielding a final target of
n=42.
Patients with S. stercoralis mono-infection who met the study criteria were counseled and provided written informed consent. Participants were treated as outpatients with ivermectin 200 µg/kg/day for 2 consecutive days. Administrative data, clinical symptoms, OD antibody titers by ELISA, eosinophil counts, and direct stool microscopy findings were recorded. Symptomatic management (e.g., antihistamines, antibiotics, liver-support medications) was provided for 5–10 days as clinically indicated. Patients were contacted by telephone during the first days of treatment to monitor adverse effects and were reassessed 1 month after treatment to evaluate clinical response. A follow-up visit at 3 months included reassessment of symptoms and repeat testing for S. stercoralis antibodies and eosinophils. Patients without symptom improvement at 1 month or experiencing adverse events were invited for earlier in-person evaluation.
Serological testing was performed using a commercial ELISA kit for
S. stercoralis infection (Viet Sinh), with reported sensitivity of 93.3% and specificity of 100%. Results were expressed as OD ratios: positive (OD ≥1), intermediate (0.8< OD <1), and negative (OD <0.8). The drug used in the study was ivermectin at a dose of 200 µg/kg/day taken 2 h before meals, for 2 consecutive days. Treatment effectiveness was evaluated according to the 2022 Vietnam Ministry of Health guidelines [
10]. At 1 month, outcomes were classified as cured (complete symptom resolution), reduced (partial symptom improvement), or not cured (no improvement). At 3 months, patients were classified as cured/recovered if symptoms resolved and OD titers were negative or decreased compared with baseline and eosinophil counts normalized (if previously elevated). Reduction was defined as partial symptom improvement or recurrence after at least 2 months with persistently positive and increased OD titers compared with baseline. Lack of improvement was classified as not cured.
Data were extracted from medical records and stored in Microsoft Excel 2019 (Microsoft Corp.). Statistical analyses were performed using IBM SPSS Statistics version 22 (IBM Corp.). Symptom changes were analyzed using the McNemar test. Changes in OD titers and eosinophil counts were assessed using the paired t-test or Wilcoxon signed-rank test, as appropriate. Treatment effectiveness was evaluated by comparing proportions of cured, reduced, and not cured cases before and after treatment using the McNemar test.
Results
During January to October 2023, the Institute’s clinic recorded 3,814 cases with seropositive ELISA tests for S. stercoralis. Of 3,814 seropositive cases for S. stercoralis, only 66 met the inclusion criteria for symptomatic mono-infection after exclusion of potential co-infections with other helminths. All 66 patients were treated with ivermectin for 2 consecutive days. At the 3-month follow-up, 42 patients completed evaluations at both 1 and 3 months.
Among the 42 patients with
S. stercoralis infection, the most common symptoms were hives/itching (88.1%), epigastric or periumbilical pain (45.2%), headache (21.4%), and loose stools (16.7%) (
Table 1). Itching without skin lesions was reported in 14/37 patients (37.8%). The typical triad (abdominal pain, urticaria, and dyspepsia/loose stools) occurred simultaneously in 2 patients (4.8%).
After 1 month of treatment, symptom persistence was uncommon: itching did not improve in 2/36 patients (5.6%), and abdominal pain did not improve in 1/17 patients (5.9%); all other symptoms resolved (
Table 2). After 3 months of treatment, itching did not improve or recurred in 21.6% of patients, and 29.4% of patients had not completely recovered from epigastric pain.
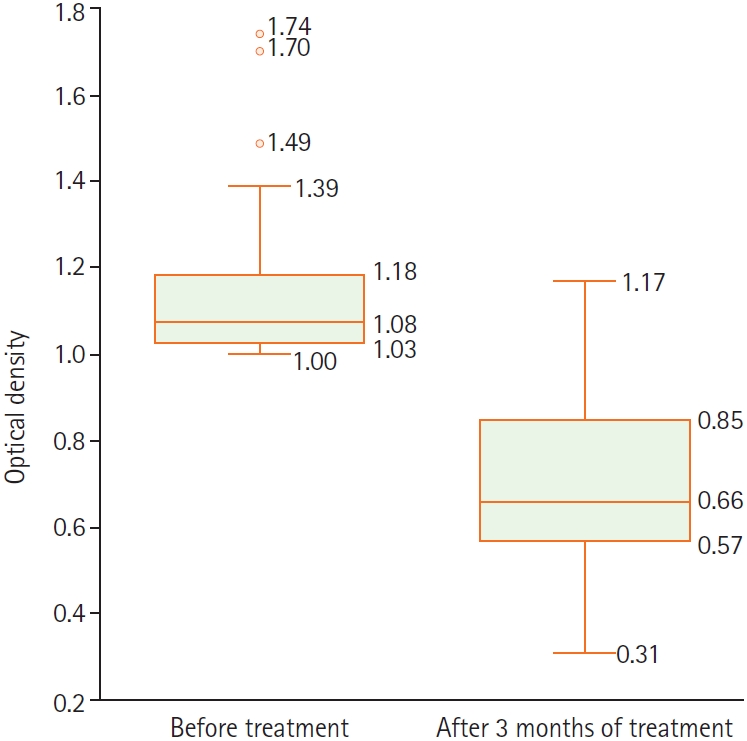
For anti–
S. stercoralis IgG, OD ratios were unevenly distributed; before treatment, OD values most commonly fell between 1.0 and <1.25, accounting for 83.3% of patients (
Table 3). OD antibody titers decreased after treatment, and this difference was statistically significant (
Fig. 1). Among the 42 patients, 5 (11.9%) had eosinophilia, defined as eosinophil count ≥0.5 G/L and eosinophil percentage >6% (
Table 3). Most eosinophilia cases improved after 3 months of treatment; however, 2 cases (5.1%) had an elevated eosinophil count at 3 months.
Based on these findings, the overall treatment effectiveness—from cure to disease reduction—of ivermectin 200 µg/kg/day for 2 consecutive days reached 95.2% at 3 months. Of the patients, 50% were completely cured, 9.5% had symptom resolution without improvement in paraclinical indices, 21.4% experienced clear symptom reduction, and 14.3% had symptom recurrence (
Table 4). Adverse effects were infrequent and included increased itching after treatment, nausea, and dry facial skin.
Discussion
Given that several helminth infections routinely screened at IMPE Quy Nhon, including
T. canis,
Fasciola spp.,
G. spinigerum, and
C. cellulosae, can present with symptoms overlapping those of
S. stercoralis infection (e.g., itching, urticaria, headache, abdominal pain), cases with co-infection or intermediate (“gray zone”) OD ratios should be excluded using ELISA IgG testing for these parasites [
11]. The follow-up period was 3 months; therefore, some patients did not attend follow-up due to distance from the study site or because symptoms resolved. Ultimately, 42 patients with
S. stercoralis mono-infection completed the study protocol.
Because patients with immunodeficiency were not included, clinical manifestations primarily involved the gastrointestinal tract and the skin/mucous membranes. The most common symptoms were itching (88.1%), urticaria (42.9%), epigastric pain (40.5%), headache (21.4%), and loose stools (16.7%) (
Table 1). These findings are consistent with common clinical presentations of strongyloidiasis, including itching, urticaria, abdominal pain, and digestive disorders/diarrhea. Symptom patterns in this study were similar to those reported elsewhere. In a study by Van De et al. [
12] of 400 patients with seropositive
S. stercoralis in northern Vietnam, the main symptoms were diarrhea (64.5%), gastrointestinal disorders (58%), epigastric pain (45.5%), itching (28%), fever (9.5%), and bloody stools (8%). In the same setting as the present study, Quang and Chuong [
9] reported epigastric pain (56.7%), nausea (34.4%), and loose stools (8.1%–24.3%) among patients with
S. stercoralis infection. Loose stools were less common (16.7%) and were generally characterized as soft but formed (approximately Bristol Stool Scale Type 5), rather than watery diarrhea, occurring 2–3 times/day without prolonged persistence. The stool was not described as fishy or foul-smelling, and outpatient stool collection was limited. Therefore, stool testing for parasites was not performed in all patients and was limited to those with digestive symptoms; results were negative in 100% of tested patients. In addition, only direct stool microscopy available at the clinic was used. Other methods (e.g., Baermann concentration or Harada–Mori culture) were not performed; therefore, treatment response was not evaluated using stool parasite clearance criteria.
Regarding symptom response, at 1 month after treatment, 22 patients (52.4%) reported complete symptom resolution, 17 (40.5%) reported partial symptom relief, and 3 (7.1%) reported no improvement (
Table 2). Two patients without improvement in urticaria received a repeat dose, and the patient with persistent abdominal pain received additional acid-suppressive therapy (a proton pump inhibitor). At 2 months, 2 patients (4.8%) still reported no symptom improvement, 6 (14.3%) experienced symptom recurrence after 2 months of resolution, 9 (21.4%) had partial symptom relief, and 25 (59.5%) reported complete symptom resolution. The most persistent symptom was itching, whereas other symptoms largely resolved after one course of treatment. Clinical improvement in this study appeared greater than that reported in other studies in Vietnam. Quang and Chuong [
9] reported symptom improvement after 3 months of 84.6% with a single dose of ivermectin and 53.33% with 2 weeks of albendazole. In the study by Vinh et al. [
13], urticaria frequently improved (51.2%) but did not completely resolve after 6 weeks of single-dose ivermectin treatment.
Anti–
S. stercoralis IgG OD ratios (
Table 3) decreased significantly by 3 months, suggesting that a decline in antibody titer may be useful for evaluating treatment response. However, 7 patients (16.7%) had persistently positive IgG titers (OD ≥0.8) despite symptom resolution. Retesting at a later interval (e.g., 6 months) may help avoid apparent false-positive results due to delayed antibody clearance or transiently increased antigen exposure after parasite death. Eosinophils were evaluated using 2 indices: absolute eosinophil count and eosinophil percentage (
Table 3). Five patients (11.9%) had elevations in both eosinophil percentage (>6%) and absolute count (≥0.5 G/L). Most cases normalized by 3 months. One patient had an eosinophil count >1.5 G/L that responded rapidly to ivermectin and returned to normal. Two patients had elevated eosinophil counts at 3 months despite clinical improvement; this may reflect transient eosinophilic responses to parasite antigens after treatment, early infection with other parasites, or other causes.
Using the treatment-effectiveness criteria, after 1 month of ivermectin 200 µg/kg/day for 2 consecutive days, 39 of 42 patients (92.9%) were classified as cured or reduced, and 3 of 42 (7.1%) were classified as not cured. At 3 months, overall effectiveness from cure to reduction was 95.2%, with 50% completely cured and 4.8% not improved. These results are comparable to, or higher than, those reported in other studies. Quang and Chuong [
9] compared single-dose ivermectin with 14 days of albendazole and reported symptom improvement rates of 84.6% at 3 months and 95.8% at 6 months for ivermectin. Vinh et al. [
13] evaluated single-dose ivermectin at 6 weeks and reported an overall effectiveness rate (reduction to cure) of 94.4%, with urticaria often reduced but not eliminated. Suputtamongkol et al. [
7] evaluated albendazole for 7 consecutive days, single-dose ivermectin, and 2 doses of ivermectin 2 weeks apart, reporting stool parasite clearance rates of 63.3% after a mean of 19 weeks, 96.8% after 39 weeks, and 93.1% after 26 weeks, respectively. Hailu et al. [
14] reported stool larval clearance of 94.6% after 2 weeks of single-dose ivermectin treatment in children aged 6–14 years, without adverse effects. Buonfrate et al. [
8] compared single-dose ivermectin with a multiple-dose regimen (200 µg/kg on days 1, 2, 15, and 16) and found symptom resolution or reduction rates at 12 months of 83% and 82%, respectively. The effectiveness of ivermectin for 2 consecutive days (95.2%) was generally within the range reported for single-dose regimens (84.6%–96.8%) [
7-
9,
13] and repeated-dose regimens (82%–93.1%) [
7,
8]. However, cure and disease-reduction criteria varied across studies, and because this study lacked a control group, no statistically significant differences between regimens can be concluded.
Case-level analysis indicated that one patient had improvement in headache but did not meet cure criteria and was found to have anemia; symptoms improved substantially after iron supplementation, and the patient was advised to consume an iron-rich diet. Two patients with persistent itchy urticaria after 1 and 3 months were switched to albendazole 400 mg × 7 days but continued to have urticaria after 6 months despite negative anti–S. stercoralis antibody results, suggesting that strongyloidiasis may have been a contributing factor rather than the sole cause; these patients were referred for evaluation of other etiologies of pruritic urticaria. Among the 42 patients, 6 (14.3%) experienced recurrence of itching after 2 months of resolution. Persistent or recurrent itching may reflect incomplete treatment response with ongoing infection, immune responses to parasite antigens after parasite death, reinfection during the 3-month monitoring period, or infection with another agent. Notably, 2 cases of recurrent itching at 3 months were retested: one was seropositive for T. canis and one was positive for C. cellulosae, despite negative screening at baseline. Epigastric pain did not completely resolve in some patients, possibly because symptoms may overlap with other gastrointestinal conditions (e.g., gastroduodenal ulcer disease, colitis) or dietary factors, which can complicate assessment of treatment response. Overall, the regimen demonstrated a favorable symptom response (92.9% cured or reduced at 1 month and 81% at 3 months). However, recurrence or persistence of symptoms—particularly itching and abdominal pain—was observed in some cases, requiring further treatment or a switch to alternative regimens. Reinfection and other contributing causes could not be excluded.
Adverse effects were infrequent and included increased itching after treatment (7.1%), nausea (4.8%), and dry facial skin (4.8%). These symptoms were transient, typically occurring within the first 1–3 days after treatment, and did not affect the treatment course.
In summary, ivermectin 200 µg/kg/day for 2 consecutive days achieved treatment effectiveness—from cure to disease reduction—of 95.2% in patients with S. stercoralis infection. Itching was the most difficult symptom to relieve. The decrease in antibody titer ratio after treatment was statistically significant (P<0.05).
Notes
-
Author contributions
Conceptualization: Tran VHT, Nguyen CD. Data curation: Tran VHT, Nguyen CD. Formal analysis: Nguyen CD. Investigation: Tran VHT, Nguyen CD, Huynh QH. Methodology: Tran VHT, Nguyen CD. Software: Tran VHT, Nguyen CD. Validation: Tran VHT, Nguyen CD. Writing – original draft: Tran VHT, Nguyen CD. Writing – review & editing: Tran VHT, Nguyen CD, Huynh QH.
-
Conflict of interest
The authors have no conflicts of interest to declare.
Fig. 1.Anti–Strongyloides stercoralis antibody titers before and after 3 months of treatment.
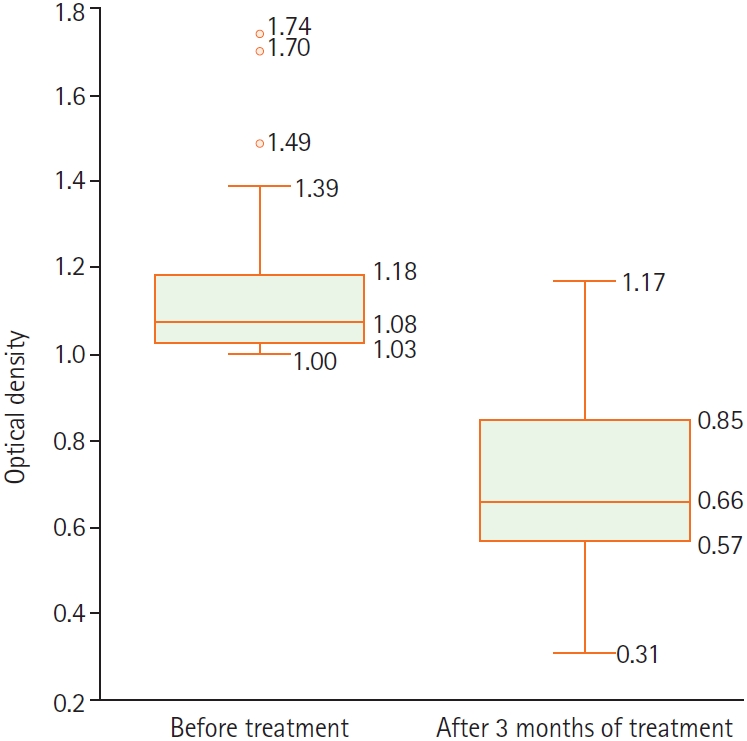
Table 1.Clinical characteristics of patients infected with Strongyloides stercoralis at IMPE Quy Nhon in 2023 (n=42)
Table 1.
|
Clinical symptom |
Value |
|
Manifestations in skin, mucosa |
|
|
Hives/itching |
37 (88.1) |
|
Urticaria |
18 (42.9) |
|
Itchy papules and inflamed tissue surrounding |
7 (16.7) |
|
Manifestations in the gastrointestinal tract |
|
|
Epigastric pain |
17 (40.5) |
|
Periumbilical pain |
4 (9.5) |
|
Loose stools |
7 (16.7) |
|
Constipation |
2 (4.8) |
|
Nausea |
4 (9.5) |
|
Manifestations related to other organs |
|
|
Headache |
9 (21.4) |
Table 2.Changes in clinical symptoms of patients infected with Strongyloides stercoralis after ivermectin treatment for 2 consecutive days at IMPE Quy Nhon in 2023
Table 2.
|
Symptom |
Before |
After 1 month |
After 3 months |
P-valuea
|
|
Resolved |
Decreased |
Not improved |
Resolved |
Decreased |
Recurred |
Not improved |
|
Manifestations in skin, mucosa |
|
Hives/itching |
36 (100) |
23 (63.9) |
11 (30.5) |
2 (5.6) |
24 (66.7) |
4 (11.1) |
6 (16.7) |
2 (5.6) |
<0.05 |
|
Urticaria |
18 (100) |
13 (72.2) |
3 (16.7) |
2 (11.1) |
12 (66.7) |
0 (0) |
4 (22.2) |
2 (11.1) |
<0.05 |
|
Itchy papules and inflamed tissue surrounding |
7 (100) |
6 (85.7) |
1 (14.3) |
0 (0) |
7 (100) |
0 (0) |
0 (0) |
0 (0) |
<0.05 |
|
Manifestations in the gastrointestinal tract |
|
Epigastric pain |
17 (100) |
11 (64.7) |
5 (29.4) |
1 (5.9) |
12 (70.6) |
5 (29.4) |
0 (0) |
0 (0) |
<0.05 |
|
Periumbilical pain |
4 (100) |
3 (75) |
1 (25) |
0 (0) |
4 (100) |
0 (0) |
0 (0) |
0 (0) |
<0.05 |
|
Loose stools |
7 (100) |
7 (100) |
0 (0) |
0 (0) |
7 (100) |
0 (0) |
0 (0) |
0 (0) |
<0.05 |
|
Constipation |
2 (100) |
2 (100) |
0 (0) |
0 (0) |
2 (100) |
0 (0) |
0 (0) |
0 (0) |
<0.05 |
|
Nausea |
4 (100) |
4 (100) |
0 (0) |
0 (0) |
4 (100) |
0 (0) |
0 (0) |
0 (0) |
<0.05 |
|
Manifestations related to other organs |
|
Headache |
9 (100) |
6 (66.7) |
3 (33.3) |
0 (0) |
8 (88.9) |
1 (11.1) |
0 (0) |
0 (0) |
<0.05 |
Table 3.Paraclinical indices before treatment and change in OD-based antibody titer after 3 months of treatment at IMPE Quy Nhon in 2023 (n=42)
Table 3.
|
Paraclinical indices |
Before treatment (n=42) |
After 3 months of treatment (n=42) |
|
Antibody titer ratio ODa
|
|
≥1.5 |
2 (4.8) |
- |
|
1.25≤ OD <1.5 |
5 (11.9) |
- |
|
1.00≤ OD <1.25 |
35 (83.3) |
4 (9.5) |
|
0.8≤ OD <1.00 |
- |
9 (21.4) |
|
<0.8 |
- |
29 (69.1) |
|
Mean±SD |
1.14±0.17 |
0.69±0.21 |
|
Median |
1.07 |
0.68 |
|
Eosinophil count (G/L)b
|
|
<0.5 |
37 (88.1) |
39 (92.9) |
|
0.5–1.5 |
4 (9.5) |
3 (7.1) |
|
>1.5 |
1 (2.4) |
0 (0) |
|
Mean of eosinophil count |
0.26±0.26 |
0.24±0.22 |
|
Eosinophil percentagec
|
|
Normal 0–6 |
35 (83.3) |
38 (90.5) |
|
Increased >6 |
7 (16.7) |
4 (9.5) |
Table 4.Therapeutic effectiveness of ivermectin 200 µg/kg/day for 2 consecutive days in patients infected with Strongyloides stercoralis after 1 and 3 months of treatment at IMPE Quy Nhon
Table 4.
|
Results of treatment (n=42) |
At 1 month |
At 3 months |
|
Overall response (cure/recovery + reduction) |
39 (92.9) |
40 (95.2) |
|
Cure/recovery |
22 (52.4) |
21 (50.0) |
|
Reduction |
17 (40.5) |
19 (45.2) |
|
Not cured |
3 (7.1) |
2 (4.8) |
|
P-valuea
|
<0.001 |
<0.001 |
References
- 1. Buonfrate D, Bisanzio D, Giorli G, et al. The global prevalence of Strongyloides stercoralis infection. Pathogens 2020;9:468. https://doi.org/10.3390/pathogens9060468
- 2. De NV, Minh PN, Bich NN, Chai JY. Seroprevalence of tissue and luminal helminths among patients in Hanoi Medical University Hospital, Vietnam, 2018. Korean J Parasitol 2020;58:387-92. https://doi.org/10.3347/kjp.2020.58.4.387
- 3. Ly DTK. Situation of patients infected with helminths examined at the Clinic of Institute of Malariology, Parasitology and Entomology Quy Nhon. J Malar Parasite Dis Control 2017;96:105-11.
- 4. Van TTH, Thuy VTT, Nhung NTC, Liem NT, Tuan PA. Current status of tissue-invading parasitic infections in patients coming for parasite screening at University Medical Center Ho Chi Minh City. Ho Chi Minh City J Med 2021;25:67-75.
- 5. World Health Organization. Strongyloidiasis [Internet]. 2022. [cited 2025 Jun 18]. Available from: https://www.who.int/teams/control-of-neglected-tropical-diseases/soil-transmitted-helminthiases/strongyloidiasis
- 6. Katzung BG. Basic and clinical pharmacology. 14th ed. McGraw-Hill Education; 2018.
- 7. Suputtamongkol Y, Premasathian N, Bhumimuang K, et al. Efficacy and safety of single and double doses of ivermectin versus 7-day high dose albendazole for chronic strongyloidiasis. PLoS Negl Trop Dis 2011;5:e1044. https://doi.org/10.1371/journal.pntd.0001044
- 8. Buonfrate D, Salas-Coronas J, Muñoz J, et al. Multiple-dose versus single-dose ivermectin for Strongyloides stercoralis infection (Strong Treat 1 to 4): a multicentre, open-label, phase 3, randomised controlled superiority trial. Lancet Infect Dis 2019;19:1181-90. https://doi.org/10.1016/S1473-3099(19)30289-0
- 9. Quang HH, Chuong NV. Comparison of the efficacy of ivermectin with albendazole in the treatment of patients infected with Strongyloides stercoralis larvae. Ho Chi Minh City J Med 2014;18:253-60.
- 10. Vietnam Ministry of Health. Decision on promulgating the guidelines for diagnosis, treatment and prevention of strongyloidiasis. Vietnam Ministry of Health; 2022.
- 11. Kolkhir P, Balakirski G, Merk HF, Olisova O, Maurer M. Chronic spontaneous urticaria and internal parasites: a systematic review. Allergy 2016;71:308-22. https://doi.org/10.1111/all.12818
- 12. Van De N, Minh PN, Van Duyet L, Mas-Coma S. Strongyloidiasis in northern Vietnam: epidemiology, clinical characteristics and molecular diagnosis of the causal agent. Parasit Vectors 2019;12:515. https://doi.org/10.1186/s13071-019-3776-1
- 13. Vinh LD, Hong TT, Du VV, Thach NK, Quang HH. Report on a series of Strongyloides stercoralis infections in the community of Duc Hoa district, Long An province and the effectiveness of treatment with a single dose of ivermectin from March 2017 to December 2017. J Malar Parasite Dis Control 2018;107:25-31.
- 14. Hailu T, Nibret E, Amor A, Munshea A, Anegagrie M. Efficacy of single dose ivermectin against Strongyloides stercoralis infection among primary school children in Amhara National Regional State. Infect Dis (Auckl) 2020;13:1178633720932544. https://doi.org/10.1177/1178633720932544