Abstract
Excretory-secretory products of Toxocara canis larvae have been considered as a major functional antigen in immune responses against toxocariasis. We studied ultrastructural localization of T. canis second-stage larval antigen using a seropositive human serum under immunogold electron microscopy. High-density gold particles were observed in the secretory cells, excretory duct, intestinal epithelium, and cuticle of the larval worm sections. The distribution of the positive reactions in the larval worms suggests that the nature of the antigen is excretory-secretory antigen including waste metabolites and secretory enzymes.
-
Key words: Toxocara canis, larva, ultrastructure, antigen, human serum
Human toxocariasis is one of the most commonly reported zoonotic helminthic infections in the world. In Korea, out of 97 healthy people with over 10% eosinophilia, 60% was positive to
Toxocara larvae excretory-secretory (TES) antigen by both immunoblot and ELISA [
1]. Also, there was a 0.9% egg positive rate of
Toxocara canis out of 662 dogs in a rural area, Korea [
2]. Three modes of pathogenesis occur in human toxocariasis. First,
T. canis larvae can cause destructive lesions that are characterized by hemorrhage and necrosis during their migration to the eye, liver, lung, brain, and heart [
3]. Second, the host immune response against migrating larvae induces inflammation and granuloma formation that is directed to the TES antigen. Third, the TES has been associated with allergy-related syndromes such as chronic urticaria, reactive arthritis, and angioderma, which suggest an allergic reaction to the TES [
4]. The excretory-secretory (ES) products of
T. canis larvae have been considered as a functional antigen in immune responses against toxocariasis because the excretory cells of the larvae contain many ES products [
5]. Then, in most immunological tests, ES recombinant antigens from
T. canis second-stage larvae have been conventionally used because the infective
Toxocara larvae are extremely difficult to detect in biopsy specimens [
6]. Some components of the TES are known to be released from the esophageal gland and lumen, i.e., midbody secretory column, which opens onto the cuticle at a secretory pore, and epicuticle under immunogold electron microscopy [
7,
8]. Although several studies on the secretory anatomy have been performed in parasitic nematode species [
5,
9,
10], there are no data on the origin of the TES itself in relation to structure and function of the excretory system. We attempted to elucidate the ultrastructural localization of the TES antigen from
T. canis second-stage larvae using a human serum that is seropositive against the TES. Determining the distribution sites of the TES will help elucidate the physiologic nature of the TES antigen.
For the primary antibody, a human serum that was serologically positive to TES by ELISA and immunoblotting analysis was used. The patient had clinical complaints of visual impairment along with floating or bubbling sensation [
11]. For a secondary antibody, goat anti-human IgG (γ-chain specific) coupled with 5 nm colloidal gold particle (Sigma, St. Louis, Missouri, USA) was used.
T. canis larvae from culture media [
12] were fixed in 2% paraformaldehyde and 0.4% glutaraldehyde in 0.2 M phosphate buffer (PB, pH 7.4) for 2 hr at room temperature, and washed with 0.2 M PB for 30 min at room temperature. The samples were dehydrated in a graded series of ethanol at 4℃ and embedded in LR gold resin (Electron Microscopy Sciences, Fort Washington, Pennsylvania, USA). The resin was polymerized at -20℃ for 2 days. An ultrathin section (90 nm) was cut and mounted on a 300-mesh nickel grid for labeling (Electron Microscopy Sciences). Sectioned specimens were incubated with drops of blocking solution (3% skim milk, 0.01% v/v Tween 20 in 0.1 M PBS) for 10 min, and incubated with a 1 : 50 dilution of the primary antibody in blocking solution for 2 hr at room temperature. The immunoreacted specimens were washed in the PBS-BSA-Tween solution (1% bovine serum albumin fraction V, 0.01% v/v Tween 20 in 0.1 M PBS), and then incubated again in a 1 : 100 dilution of conjugate in PBS-BSA-Tween at 4℃ overnight. They were washed again in PBS-Tween (0.01% v/v Tween 20 in 0.1 M PBS) with 3 changes for 10 min and postfixed with 2.5% glutaradehyde in 0.1 M PB (pH 7.4) for 15 min at room temperature. After washing with 0.1 M PBS, the specimens were dried by saturating the filter paper, negatively stained with 2% w/v uranyl acetate in 50% methanol for 25 min at 37℃ while protected from light, and washed with distilled water. The specimens were stained with Reynold's lead citrate solution (1.3 g lead nitrate and 1.76 g sodium citrate in 30 ml distilled water) at room temperature for 5 min and washed with distilled water. The grid was examined under a transmission electron microscope (1200 EXCII; JEOL, Tokyo, Japan).
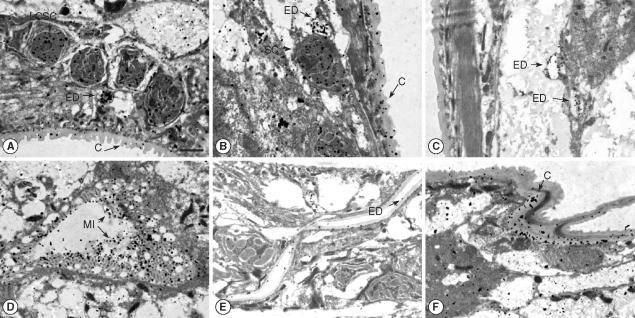
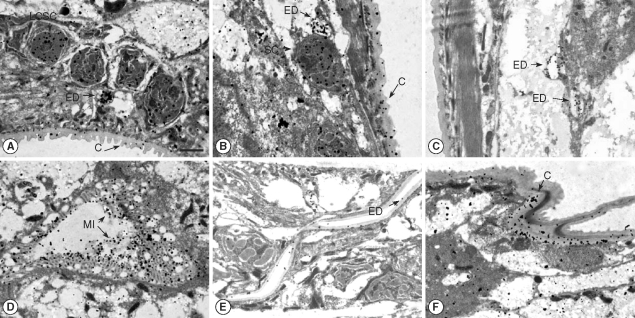
T. canis larvae have a large esophagus with an associated dorsal esophageal gland and a secretory system composed of large columnar secretory cells that are ducted to excretory pores and opening onto the cuticle. The intestine at this infective larval stage is closed and presumed to be functional. The immunogold particles were distributed in the large column-like excretory cells (
Fig. 1A), excretory ducts (
Fig. 1A-C, E), secretory cells (
Fig. 1B), microvilli of the intestine (
Fig. 1D), and cuticle (
Fig. 1A, B, F).
In general, the excretory systems of nematode species exhibit extremely variable morphologies. This variability suggests that the system has different functions in different species and may serve multiple functions in some nematodes such as osmoregulation, waste-product clearance, secretion, and export of some enzymes. Gland cells are associated with the excretory system in many species, and it has been suggested that secreted products may be released to the outside environment through the excretory duct or reach other internal tissues via excretory canals [
10]. In previous
T. canis immunogold studies, it was found that the esophageal gland and lumen are strongly reactive with monoclonals Tcn-4, Tcn-5, and Tcn-8, and because the posterior portion of the gut is closed, the products of this gland are released through the oral aperture. A distinct anti-TES antibody (Tcn-2) localizes solely to the midbody secretory column, which opens onto the cuticle at a secretory pore. Thus, the secretory apparatus is probably functional in this stage of the parasite as an important source of TES products [
7,
8]. The surface-labeled TES-70 glycoprotein is linked with a more hydrophobic stratum at the surface, and a prominent 32-kDa glycoprotein, TES-32, is more strongly represented within the cuticle itself. An extracuticular surface coat has been detected on
Toxocara larvae prior to hatching from the egg and during all stages of in vitro culture, suggesting that it may play a role both in protecting the parasite on hatching in the gastrointestinal tract and on subsequent tissue invasion by evading host immune responses directed at surface antigens [
8]. Those 2 studies were conducted using monoclonal antibodies against the TES.
Kondo et al. [
5] reported that they unclearly showed the excretory cells of
T. canis larvae by immunoelectron microscopy using rabbit sera, and also there are no data about the origin of TES antigen using human sera. In the present study, a human serum detected by immunodiagnosis was used and the origin of the antigen was detected more clearly. In a negative control omitting the first antibody, the control section stained with sera did not react to the TES antigen (data not shown). Recent studies suggested that the toxocariasis frequently assumes various clinical features such as chronic weakness, abdominal pain, allergy, hypereosinophilia, and various neurological syndromes in humans [
5,
13,
14]. However, although TES may be involved with these clinical symptoms, the present study still remains unsolved because serological assays can give false positive reactions due to cross reactions with other parasitic helminths commonly found in tropical areas [
6]. For a more clear understanding about the source of the TES antigen, we labeled colloidal gold particles using a human serum. Our results are supported by those obtained previously by Kondo et al. [
5]. In other nematode immunogold studies using electron microscopy, the glycoprotein (GP31) antigen has been located in secretory organelles within the cells of the esophageal glands in sheep nematode (
Ostertagia circumcincta) larvae [
15]. The monoclonal antibodies against excretory-secretory-metabolic antigens of filarial parasites were found to label the cuticular portions of third-stage larvae and adults of lymphatic parasites but not the epicuticular surface according to immunoelectron microscopic studies [
16].
Based on the above findings, it is suggested that the source of the ES antigen from the larvae of T. canis might be produced or excreted from secretory granules. The antigen would be released from the esophageal glands of the worm via the esophagus to the os or via the secretory duct to the outside of the cuticle. In addition, substances may be excreted from the hypodermis directly through the cuticle, which is water permeable in all stages. From these findings, we suggest that the nature of the antigen of T. canis larvae might be as a secretory enzyme. The release of surface antigens may also provide an important clue to explain how larvae evade the host's immune response in human toxocariasis.
ACKNOWLEDGEMENTS
This work was supported by a Korea Research Foundation Grant (2003-015-E00113).
References
Fig. 1Immunogold findings of Toxocara canis larva reacted with a seropositive human serum. Bar = 1 µm. Arrows indicate each particle. (A) High density immunogold particles are distributed in the large column-like secretory cell (LCSC), excretory duct (ED), and cuticle (C); (B) High density immunogold particles are shown in the secretory cell (SC), excretory duct (ED), and cuticle; (C) Immunogold particles are distributed in the excretory duct (ED); (D) High dense particles are displayed in the microvilli of the intestine (MI); (E) Immunogold particles are shown in the excretory duct; (F) High density immunogold particles are distributed in the cuticle (C).
Citations
Citations to this article as recorded by

- Toxocara canis-originated recombinant C-type lectin improves the disability scores of experimental autoimmune encephalomyelitis in murine in vivo models
Mahsa Shahbakhsh, Fateme Jalousian, Seyed Hossein Hosseini, Abdorreza Naser Moghadasi, Parviz Shayan, Samad Farashi Bonab, Parmida Malekzade, Mohammad Vojgani, Mahya Lalehpour
Journal of Neuroimmunology.2025; 402: 578569. CrossRef - Producción y evaluación del antígeno recombinante Tes-30 de Toxocara canis para el inmunodiagnóstico de toxocariasis
Ana M. Olave, Jairo A. Mesa, Jorge H. Botero, Edwin B. Patiño, Gisela M. García, Juan F. Alzate
Biomédica.2015;[Epub] CrossRef - Evaluación de un antígeno purificado para el diagnóstico de toxocariosis
Graciela Santillán, Vanesa Bastin, Graciela Céspedes, Adriana Monkiewicz
Revista Argentina de Microbiología.2013; 45(2): 80. CrossRef - Frequency of unexpected antibody and consideration during transfusion
Ki-Ho Ko, Byung-Hoon Yoo, Kye-Min Kim, Woo-Yong Lee, Jun-Heum Yon, Ki-Hyuk Hong, Tae-Hee Han
Korean Journal of Anesthesiology.2012; 62(5): 412. CrossRef