Abstract
Kudoa septempunctata, a myxozoan, has been identified as the causative agent of foodborne illnesses associated with the consumption of raw olive flounder. To develop effective control methods against this parasite, fundamental research—including viability determination, transcriptome analysis, and antigenicity assessment of K. septempunctata myxospores—is required. This research necessitates the purification of the parasites. Sequential trypsin digestion, followed by density gradient purification, was performed to isolate the K. septempunctata myxospores. Further purification was achieved through fluorescence-activated cell sorting at a concentration of 106 to 107 myxospores/ml. The results demonstrated that the combination of trypsin digestion and density gradient methods consistently produced approximately 40 times more viable myxospores than the density gradient method alone. Additionally, the fluorescence-activated cell sorting method enhanced the purity of the myxospores by approximately 10%. The procedures described in this study will support research (such as RNA-sequencing, proteomics, vaccine antigen preparation) aimed at developing control methods for K. septempunctata including the fundamental research.
-
Key words: Kudoa septempunctata, purification, trypsin, fluorescence-activated cell sorting
Myxozoans are tiny parasites belonging to the phylum Cnidaria and include over 2,000 species [
1]. These parasites have a complete life cycle involving both vertebrate and invertebrate hosts, with myxospores produced in vertebrates and actinospores in invertebrates [
1]. While most myxozoans do not pose a serious threat to their hosts, some can cause mass mortality, myoliquefaction, and foodborne illnesses [
2-
4].
Kudoa septempunctata (family: Kudoidae) was first reported in cultured olive flounder exported from Korea to Japan and has been identified as a causative agent of foodborne illness associated with the consumption of the raw olive flounder
Paralichthys olivaceus [
2,
5]. The olive flounder is the most widely cultured fish species in Korea. However, foodborne illnesses associated with
K. septempunctata are reported annually, and quarantine screening for
K. septempunctata has contributed to a decline in olive flounder exports to Japan. Various studies have been conducted to reveal the mechanisms of foodborne illnesses caused by
K. septempunctata and have evaluated the effects of various chemicals on their viability [
6-
9]. Traditionally, myxospores of
K. septempunctata have been purified by Percoll density gradient centrifugation [
7]. However, myxospores purified from fish muscle by the Percoll method are often contaminated with debris such as host cells, erythrocytes, and pseudocysts. Additionally, the efficiency of the Percoll method may not be consistent depending on the individual. This inconsistency primarily arises during the homogenization of fish muscle, as it can be challenging to maintain a consistent force to effectively separate pseudocysts and myxospores. Consequently, if contaminants with a similar density to myxospores are produced, they may coalesce in the pellet with the myxospores. Such contamination and variability in purification efficiency can interfere with molecular, biological, and biochemical analyses. Trypsin digestion has been used to isolate parasites from host tissue [
10,
11], and fluorescence-activated cell sorting (FACS) can separate particles in heterogeneous suspensions using optical signals, including light scatter and fluorescence. In a label-free setting, forward scatter (FSC) is primarily influenced by particle size, while side scatter (SSC) reflects optical heterogeneity and internal structural complexity; therefore, FSC/SSC gating can be used to enrich morphologically uniform targets such as myxospores while excluding other host-derived contaminants [
12]. The present study aimed to purify the myxospores of
K. septempunctata using a combination of trypsin digestion, density gradient centrifugation and FACS, and to evaluate the effectiveness of this combined approach.
Olive flounder (
n=10; mean total length, 33.6 cm; mean body weight, 439.4 g) were obtained from an olive flounder farm that experienced annual
K. septempunctata infection. As the fish were dead, the experiment did not require Institutional Animal Care and Use Committee approval. Squash preparations of trunk muscles were examined microscopically to detect
K. septempunctata pseudocysts. Pseudocysts were found in 5 fish, and the olive flounder with the highest number of pseudocysts was selected for the experimental sample. The myxospores within the pseudocysts were identified as
K. septempunctata based on their morphological characteristics, which included an asymmetrically stellate shape with 6–7 shell valves (mainly 7) and polar capsules. Six trunk muscle samples (1 g each), each containing hundreds of pseudocysts per gram, were collected. Three muscle samples were homogenized with phosphate buffered saline (PBS) on test sieve (200 μm, Chunggye Sieve), and myxospores were then isolated using Percoll (Sigma-Aldrich) density gradient centrifugation as previously described [
10]. The other 3 samples were minced and mixed with 10 ml of PBS containing 10 mg of trypsin (1:250, Amresco) and 100 µl of antibiotics (10,000 µg/ml of streptomycin and 10,000 U/ml of penicillin, Gibco). The samples were incubated in a shaking incubator at 25℃ and 180 rpm for 2 h. The resultant slurry was homogenized with PBS on the test sieve (200 μm), and myxospores were isolated using the Percoll method. The number of isolated myxospores was determined under a microscope using a hemocytometer. To determine the viability of the isolated myxospores, spores (5.3×10
5 spores/900 μl) were treated with 100 μl of PBS (live spores) and 10% formalin (dead spores) for 30 min, respectively. A total of 10,000 spores were analyzed, and the proportion of live and dead spores were determined using a flow cytometer (CytoFlex), following the protocol established by Shin et al. [
8]. The viability test was conducted in triplicate to ensure reliability. The isolated myxospore samples were adjusted to 3 concentrations: 1.0×10
7 spores/ml, 5.0×10
6 spores/ml, and 1.0×10
6 spores/ml. These samples were sorted into 3 groups (A, B, and C) based on differences in FSC and SSC using FACS (CytoFLEX SRT, Beckman Coulter). After sorting, spores from each group were examined microscopically to confirm the morphological characteristics. The sorted myxospores were then concentrated back to their original concentrations, and the purity of the myxospores before and after sorting was evaluated using a flow cytometer. Statistical analyses were conducted using IBM SPSS Statistics version 30 (IBM Corp.). An independent Student’s
t-test was employed to compare the number of myxospores purified by a combination of trypsin digestion and Percoll versus Percoll alone. Differences were considered significant at
P<0.05.
The purification efficiencies of the methods evaluated in this study are shown in
Figs. 1 and
2, and
Table 1. Isolation of
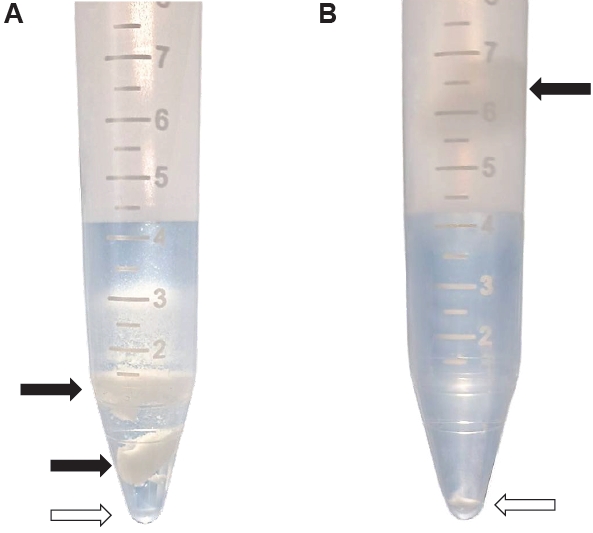
K. septempunctata myxospores using the combination of trypsin digestion and Percoll solution resulted in distinct separation of myxospores and fish muscle compared to Percoll alone (
Fig. 1). The mean number of myxospores isolated by the trypsin and Percoll method was 3.8×10
6 spores/g, whereas only 9.7×10
4 spores/g were obtained using Percoll alone (
Table 1). The
P-value was statistically significant at 0.006 for the difference in purification that depends on the purification methods. The viability of myxospores isolated using the combination method was approximately 90%.
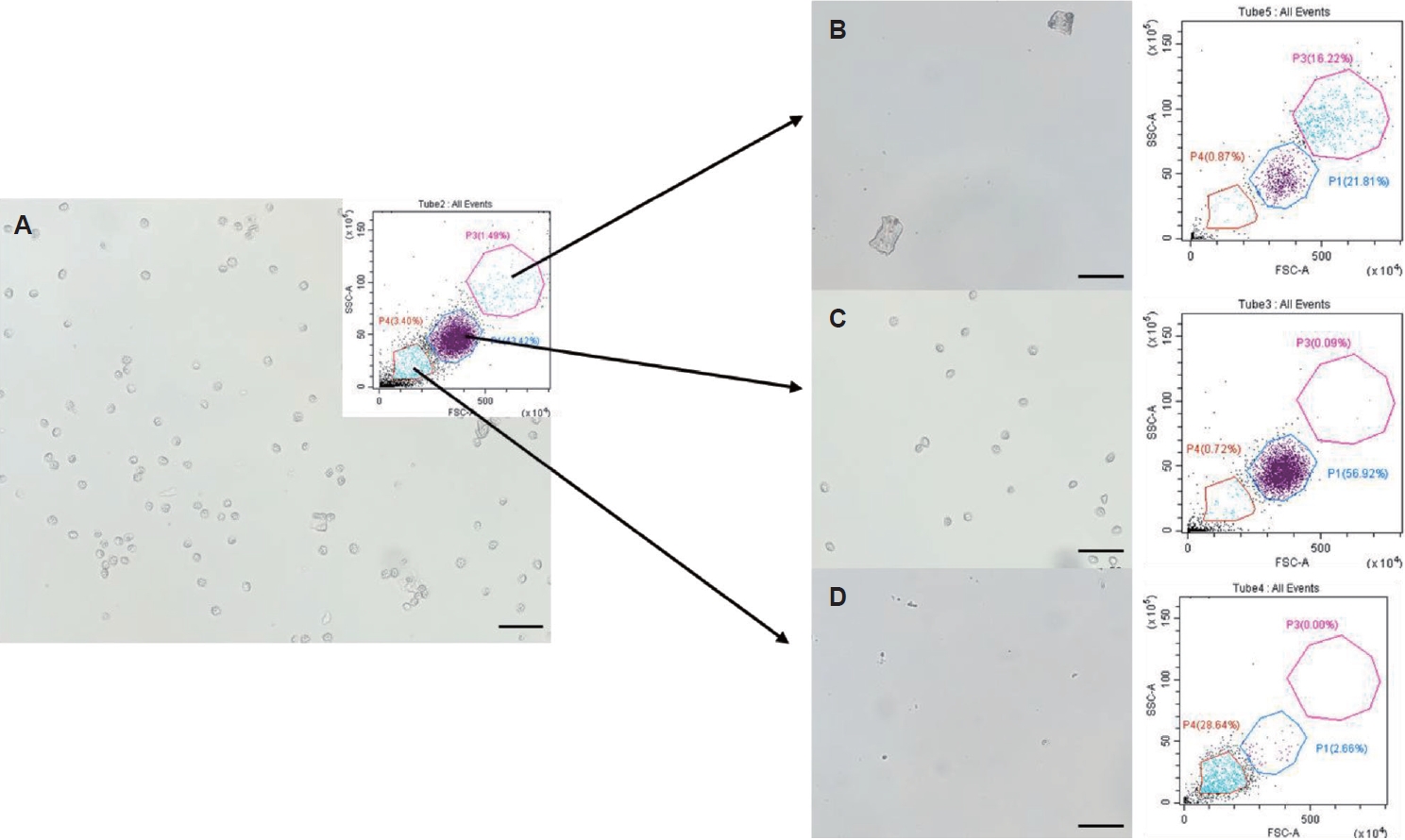
In the CytoFLEX SRT configuration (
Fig. 2; FSC-A in ×10
4 and SSC-A in ×10
5, shown in arbitrary instrument units), intact
K. septempunctata myxospores formed a dominant cluster within an intermediate-scatter window (Gate P1; approximately FSC-A ~250–450 and SSC-A ~20–70). By contrast, events with higher FSC and SSC (Gate P3; approximately FSC-A ~450–800 and SSC-A ~70–140) were enriched for larger irregular structures consistent with fragments of pseudocysts and low-scatter events (Gate P4; approximately FSC-A ≤200 and SSC-A ≤30) primarily represented small particulate debris from muscle. Importantly, these classifications were supported by microscopy of the sorted fractions: the medium-scatter fraction contained the purest myxospore preparation (
Fig. 2A,
C), whereas the high- and low-scatter fractions contained large particles and small debris, respectively (
Fig. 2A,
B,
D). Collectively, this scatter-based FACS step improved myxospore purity by 8.5%–11.6% across samples (
Table 2), providing a practical additional cleanup step after trypsin digestion and density gradient purification.
The isolation and purification of myxospores are crucial for the execution of the fundamental research on myxozoans. Although gradient density purification by Percoll was suggested by previous studies [
6-
9], it is limited to myxospores and does not employ vegetative stages, and the results may vary depending on the individual performing the procedure. Previous studies have reported the isolation and purification of parasites by trypsin digestion, which showed high recovery rates for
Toxoplasma gondii tachyzoites and
Myxobolus (formerly
Myxoma)
cerebralis myxospores [
10,
11]. However, trypsin digestion can negatively affect the viability of some parasites. Wu et al. [
11] showed that more than 95% of
T. gondii remained viable after purification with a 3-μm filter or CF-11 cellulose or Percoll. In contrast, trypsin digestion resulted in viability rates of 65% and 75%. Furthermore, Ohnishi et al. [
6] reported that trypsin affects the release of sporoplasms from myxospores of
K. septempunctata, indicating that the myxospores were no longer viable. In contrast, the combination of trypsin and Percoll methods in the present study demonstrated a distinct separation of
K. septempunctata myxospores from fish muscle, consistently yielding higher number of myxospores, compared to the Percoll method alone. The myxospores exhibited approximately 90% viability, and the release of sporoplasm by trypsin was not observed, which is consistent with the results of a previous study [
7]. Even though we employed trypsin on the myxospores with a higher concentration (1 mg versus 400 μg/ml), for a longer time (2 h versus 1 h), and at a similar temperature (25℃ versus room temperature) compared to the Ohnishi experiment, these conditions did not affect sporoplasm release. Furthermore, Shin et al. [
7] demonstrated that sporoplasm release was affected by glucose uptake and metalloprotease in the myxospores rather than by trypsin. Therefore, trypsin can be utilized for the purification of
K. septempunctata myxospores without affecting their viability. Although the precise reason for the improved isolation efficiency remains unclear, it is hypothesized that this phenomenon may occur because muscle fibers can be separated into more consistent sizes compared to manual homogenization.
FACS is a laser-based technique used to isolate and analyze individual cells from heterogeneous mixtures based on their physical and fluorescence characteristics. This method has been applied for the isolation of specific cells (such as immune, stem, and cancer cells) and the detection of pathogenic bacteria [
13-
15]. Previous studies have also reported the use of FACS for the isolation of parasites or cells infected by parasites [
16,
17]. In the present study, myxospores of
K. septempunctata were isolated using a combination of trypsin and Percoll and then further purified by FACS based on their FSC and SSC values. The purity of the myxospores improved by approximately 10% across the 3 samples tested whereas the total number of myxospores decreased by approximately 20–30 (data not shown). FSC is generally proportional to the cell-surface area or size, while SSC is proportional to cell granularity or internal complexity. Our results showed that Group A, characterized by high FSC and SSC values, primarily contained large debris and pseudocysts. This suggests that the intact pseudocysts or aggregated muscle fibers possess a large cross-sectional area and higher structural complexity than individual myxospores. In contrast, Group C, with low FSC and SSC values, consisted of fine muscle debris and fragmented tissues smaller than the myxospores. However, reliance solely on FSC and SSC parameters resulted in a trade-off between purity and yield. Furthermore, FSC/SSC-only sorting may show variable performance depending on instrument configuration and sample characteristics, which should be carefully considered when applying this method under different experimental conditions. Therefore, there are several considerations when applying FACS to purify myxospores of the parasites. First, applying pulse-geometry gates (e.g., FSC-A vs. FSC-H and/or FSC/SSC width) to exclude aggregates/doublets, which is a common strategy to reduce artifactual events and improve purity in flow cytometry and sorting [
18]. Second, using a target-specific fluorescent marker. The overlap between myxospores and host-derived debris cannot be completely eliminated, particularly when myxospores aggregate or attach to tissue fragments. This likely contributes to the observation that some myxospores appeared in the high-scatter fraction and to the overall loss in recovery after sorting. Therefore, the application of a myxospore-specific fluorescent marker in FACS can help mitigate this limitation. Previous studies have reported a higher purity of specific cells when FACS is conducted with antibody-conjugated fluorescence [
19,
20]. Choi et al. [
19] demonstrated higher purity of lymphocytes using CD45 (antibody-conjugated fluorescence)/SSC gating in FACS compared with FSC/SSC gating. Liao et al. [
20] successfully purified a rare population of immune cells, specifically plasmacytoid dendritic cells, from the bone marrow of mice using FACS and various fluorescent dyes and antibodies. Lastly, although this study focused on physical separation to maintain the native state of spores, the use of viability dyes (e.g., propidium iodide or SYTO 9) in conjunction with FACS would allow for the simultaneous purification and viability assessment of myxospores.
In conclusion, the sequential purification used in this study (a combination of trypsin and Percoll, and the utilization of FACS) consistently increases the purity of the myxospores of K. septempunctata. However, this study has limitations that did not identify the effect of FACS on the viability of myxospores, and FACS can cause more than a 20% loss. To further improve purity while reducing loss, future work should evaluate the combination of FSC/SSC gating with pulse geometry and antibodies that specifically target the myxospores and validated viability dyes.
Notes
-
Author contributions
Conceptualization: Hong HK, Park Y, Shin SP. Funding acquisition: Shin SP. Methodology: Hong HK, Park Y, Shin SP. Project administration: Shin SP. Supervision: Shin SP. Validation: Park Y. Writing – original draft: Hong HK. Writing – review & editing: Park Y, Shin SP.
-
Conflict of interest
The authors have no conflicts of interest to declare.
-
Funding
This study was supported by a research grant from Kongju National University Industry-University cooperation foundation in 2025 and the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Korean government (MSIT) (RS-20220NR069562).
Fig. 1.Isolation of Kudoa septempunctata myxospores from the trunk muscle of olive flounder. (A) Isolation using density gradient centrifugation. (B) Isolation using trypsin digestion followed by density gradient centrifugation. Black arrows indicate fish muscle, and empty arrows indicate the pellet of myxospores.
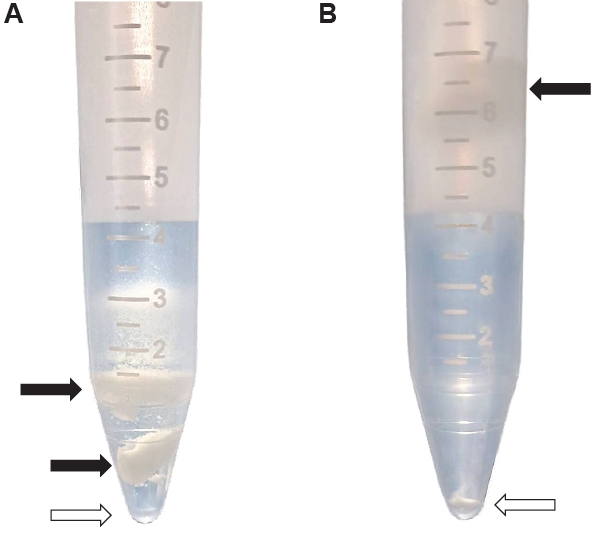
Fig. 2.Purification of Kudoa septempunctata myxospores using fluorescence-activated cell sorting. (A) Sample prior to sorting. (B) Sorted with high forward scatter (FSC) and side scatter (SSC) values. (C) Sorted with medium FSC and SSC values. (D) Sorted with low FSC and SSC values. Scale bar=50 μm.
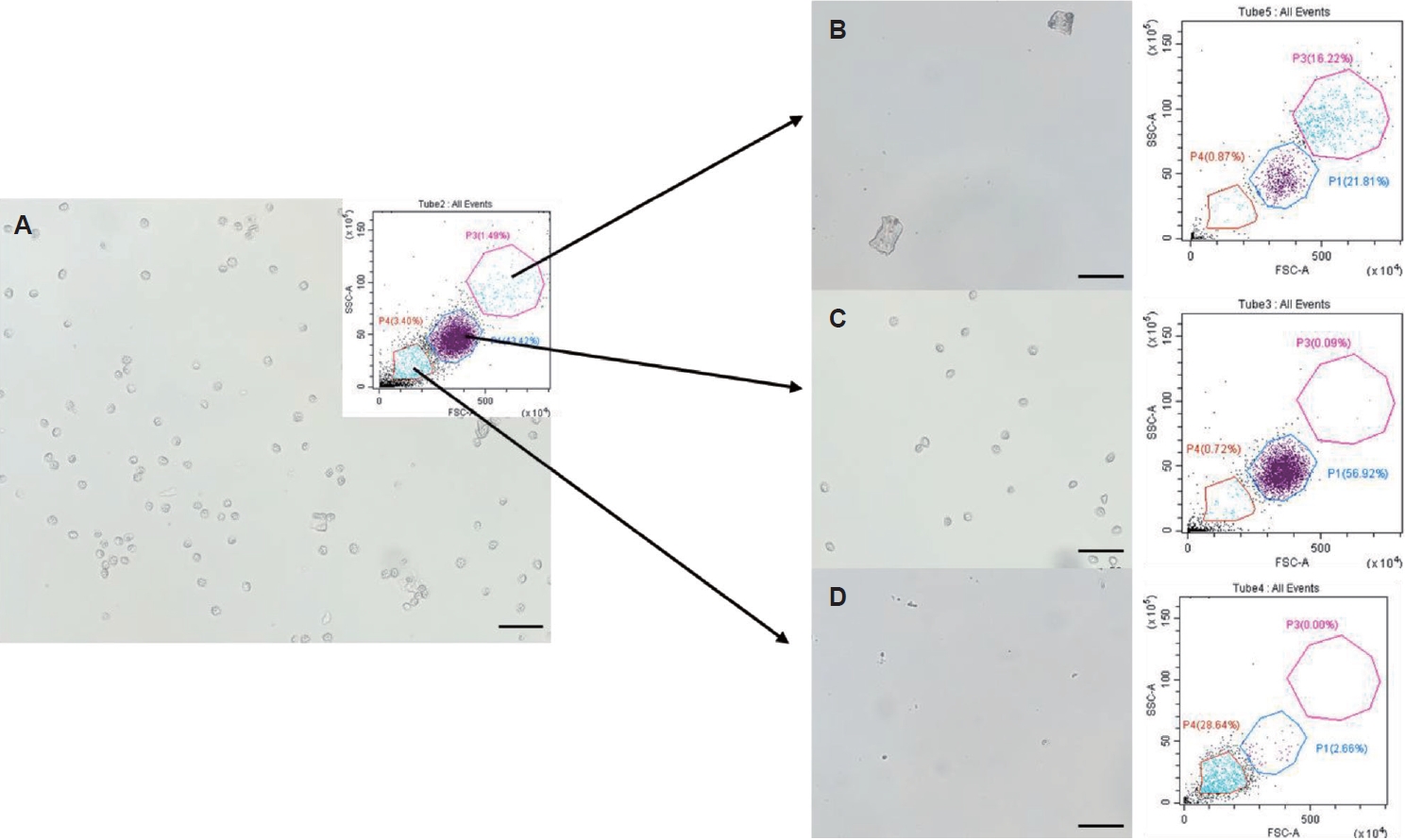
Table 1.Efficiencies of isolation methods of Kudoa septempunctata from olive flounder muscle
Table 1.
|
Isolation methods |
Samples |
No. of isolated myxospores/g |
Mean No. of isolated myxospores/g |
|
Trypsin and Percoll |
1 |
3.6×106
|
3.8×106
|
|
2 |
4.4×106
|
|
3 |
3.4×106
|
|
Percoll |
1 |
2.0×105
|
9.7×104
|
|
2 |
6.0×104
|
|
3 |
3.0×104
|
Table 2.Purity change of Kudoa septempunctata myxospores sorted by fluorescence activated cell sorting
Table 2.
|
Parasite concentration (spores/ml) |
Purity of unsorted K. septempunctata
|
Purity of sorted K. septempunctata
|
|
1.0×107
|
87.8 |
96.9 |
|
5.0×106
|
87.3 |
95.8 |
|
1.0×106
|
86.5 |
98.1 |
References
- 1. Lom J, Dykova I, Dyková I. Myxozoan genera: definition and notes on taxonomy, life-cycle terminology and pathogenic species. Folia Parasitol 2006;53:1-36. https://doi.org/10.14411/fp.2006.001
- 2. Kawai T, Sekizuka T, Yahata Y, et al. Identification of Kudoa septempunctata as the causative agent of novel food poisoning outbreaks in Japan by consumption of Paralichthys olivaceus in raw fish. Clin Infect Dis 2012;54:1046-52. https://doi.org/10.1093/cid/cir1040
- 3. Yanagida T, Palenzuela O, Hirae T, et al. Myxosporean emaciation disease of cultured red sea bream Pagrus major and spotted knifejaw Oplegnathus punctatus. Fish Pathol 2008;43:45-8. https://doi.org/10.3147/jsfp.43.45
- 4. Yokoyama H, Whipps CM, Kent ML, Mizuno K, Kawakami H. Kudoa thyrsites from Japanese flounder and Kudoa lateolabracis n. sp. from Chinese sea bass: causative myxozoans of post-mortem myoliquefaction. Fish Pathol 2004;39:79-85. https://doi.org/10.3147/jsfp.39.79
- 5. Matsukane Y, Sato H, Tanaka S, Kamata Y, Sugita-Konishi Y. Kudoa septempunctata n. sp. (Myxosporea: Multivalvulida) from an aquacultured olive flounder (Paralichthys olivaceus) imported from Korea. Parasitol Res 2010;107:865-72. https://doi.org/10.1007/s00436-010-1941-8
- 6. Ohnishi T, Kikuchi Y, Furusawa H, Kamata Y, Sugita-Konishi Y. Kudoa septempunctata invasion increases the permeability of human intestinal epithelial monolayer. Foodborne Pathog Dis 2013;10:137-42. https://doi.org/10.1089/fpd.2012.1294
- 7. Shin SP, Zenke K, Yokoyama H, Yoshinaga T. Factors affecting sporoplasm release in Kudoa septempunctata. Parasitol Res 2015;114:795-9. https://doi.org/10.1007/s00436-014-4305-y
- 8. Shin SP, Hong HK, Jin CN, et al. Effect of epigallocatechin gallate on viability of Kudoa septempunctata. Korean J Parasitol 2020;58:593-7. https://doi.org/10.3347/kjp.2020.58.5.593
- 9. Yokoyama H, Funaguma N, Kobayashi S. In vitro inactivation of Kudoa septempunctata spores infecting the muscle of olive flounder Paralichthys olivaceus. Foodborne Pathog Dis 2016;13:21-7. https://doi.org/10.1089/fpd.2015.2003
- 10. Markiw ME, Wolf K. Myxosoma cerebralis: isolation and concentration from fish skeletal elements: sequential enzymatic digestions and purification by differential centrifugation. J Fish Res Board Can 1974;31:15-20. https://doi.org/10.1139/f74-003
- 11. Wu L, Chen SX, Jiang XG, et al. Separation and purification of Toxoplasma gondii tachyzoites from in vitro and in vivo culture systems. Exp Parasitol 2012;130:91-4. https://doi.org/10.1016/j.exppara.2011.10.006
- 12. Cossarizza A, Chang HD, Radbruch A, et al. Guidelines for the use of flow cytometry and cell sorting in immunological studies (second edition). Eur J Immunol 2019;49:1457-973. https://doi.org/10.1002/eji.201970107
- 13. Fei C, Nie L, Zhang J, Chen J. Potential applications of fluorescence-activated cell sorting (FACS) and droplet-based microfluidics in promoting the discovery of specific antibodies for characterizations of fish immune cells. Front Immunol 2021;12:771231. https://doi.org/10.3389/fimmu.2021.771231
- 14. Mayol JF, Loeuillet C, Hérodin F, Wion D. Characterisation of normal and cancer stem cells: one experimental paradigm for two kinds of stem cells. Bioessays 2009;31:993-1001. https://doi.org/10.1002/bies.200900041
- 15. Moon J, Kim G, Lee S, Park S. Identification of Salmonella Typhimurium-specific DNA aptamers developed using whole-cell SELEX and FACS analysis. J Microbiol Methods 2013;95:162-6. https://doi.org/10.1016/j.mimet.2013.08.005
- 16. Einarsson E, Lassadi I, Zielinski J, et al. Development of the myzozoan aquatic parasite Perkinsus marinus as a versatile experimental genetic model organism. Protist 2021;172:125830. https://doi.org/10.1016/j.protis.2021.125830
- 17. Linares M, Viera S, Crespo B, et al. Identifying rapidly parasiticidal anti-malarial drugs using a simple and reliable in vitro parasite viability fast assay. Malar J 2015;14:441. https://doi.org/10.1186/s12936-015-0962-2
- 18. Stadinski BD, Huseby ES. How to prevent yourself from seeing double. Cytometry A 2020;97:1102-4. https://doi.org/10.1002/cyto.a.24045
- 19. Choi YH, Shim H, Park CJ, et al. Flow cytometric assays for lymphocyte subset enumeration: CD45 is inevitable for lymphocyte gating and CD16 is essential for NK cells. Lab Med Online 2013;3:79-87. https://doi.org/10.3343/lmo.2013.3.2.79
- 20. Liao X, Makris M, Luo XM. Fluorescence-activated cell sorting for purification of plasmacytoid dendritic cells from the mouse bone marrow. J Vis Exp 2016;4:54641. https://doi.org/10.3791/54641