Abstract
This study aimed to investigate the role of palmitic acid (PA) in modulating the neutrophil response during Trichomonas vaginalis infection. A human neutrophil-like cell model (nHL-60) was established by differentiating the human promyelocytic leukemia cell line HL-60 with 1.3% dimethyl sulfoxide. The expressions of the genes IL-8, NF-κB, MyD88, and TLR4 and the production of reactive oxygen species (ROS) of differentiated cells were compared, using end-point RT-PCR and nitroblue tetrazolium reduction, in the presence or absence of 1 mM PA and 100 μM of docosahexaenoic acid. Additionally, oxidative burst activity was also evaluated in response to T. vaginalis stimulation in the presence of either PA or docosahexaenoic acid. nHL-60 cells showed increased expression of key mediators of the TLR4 signaling pathway, and enhanced ROS production upon phorbol 12-myristate 13-acetate stimulation. Peripheral blood neutrophils exposed to T. vaginalis in the presence of PA exhibited higher ROS production than neutrophils stimulated by the parasite only. These findings indicate that PA enhances neutrophil activation to a secondary stimulus with T. vaginalis. They provide new insights into the role of metabolic factors in host-pathogen interactions.
-
Key words: Neutrophils, palmitic acid, Trichomonas vaginalis, reactive oxygen species, Toll-like receptor 4
Fatty acids are crucial modulators of immune responses, particularly saturated fatty acids, which often induce pro-inflammatory and immunomodulatory effects. Palmitic acid (PA), the most abundant saturated fatty acid in human plasma, has been shown to activate immune cells by inducing cytokine production, reactive oxygen species (ROS) generation, and cell migration [
1]. PA induces immune activation through the Toll-like receptor 4 (TLR4) signaling cascade, triggering inflammatory responses in macrophages and other immune cells [
2,
3]. These effects contribute to both protective and pathological immune functions depending on the activation context [
4,
5].
Neutrophils are vital components of the innate immune system, acting as the first line of defense against invading pathogens. They contribute to eliminate microorganisms through mechanisms such as phagocytosis, degranulation, and the release of ROS [
6]. Pattern recognition receptors, including TLR4, tightly regulate neutrophil activation by binding microbial and non-microbial components, initiating several inflammatory pathways [
7]. PA mediates macrophage activation through TLR4 [
8]. However, its effects on human neutrophils have remained underexplored with conflicting reports regarding its role in ROS production and inflammatory signaling [
9,
10]. Evidence suggests that PA can modulate neutrophil function by enhancing their migration and activation [
1], yet excessive or dysregulated exposure to PA may lead to aberrant signaling through TLR4, highlighting the need for further studies to clarify the mechanisms involved.
Trichomoniasis is a widespread sexually transmitted infection caused by the parasite
Trichomonas vaginalis. This infection correlates with increased susceptibility to human immunodeficiency virus, infertility, and cervical cancer [
11,
12].
T. vaginalis triggers a robust inflammatory response, leading to the infiltration of neutrophils into the vaginal mucosa. Neutrophils contribute to parasite elimination by releasing ROS, cytokines, IL-8, and other chemokines that regulate immune cell recruitment and the inflammatory response [
13].
It is unknown if PA directly modulates neutrophil activity during trichomoniasis and whether this interaction has beneficial or detrimental consequences for pathogen elimination. To address this gap, we investigated the role of PA in modulating human neutrophil responses to T. vaginalis infection.
The human promyelocytic leukemia cell line HL-60 (American Type Culture Collection, CCL-240) was cultured in Dulbecco's modified eagle medium and differentiated into a neutrophil-like phenotype (nHL-60) by incubation with 1.3% dimethyl sulfoxide (DMSO) for 6 days. The morphological and functional characteristics of nHL-60 were compared to those of peripheral blood neutrophils isolated from healthy donors by density gradient centrifugation using Histopaque; no RBC lysis was performed. The production of ROS, was measured by incubating cells with 1 mg/mL nitroblue tetrazolium (NBT) at 37°C for 30 min, using the cells with 500 nM phorbol 12-myristate 13-acetate (PMA) as a positive control. The resulting formazan crystals were analyzed using optical microscopy, dissolved in 2 M KOH y DMSO and quantified spectrophotometrically at 620 nm. Additionally, cells were incubated with 100 ng/ml lipopolysaccharide (LPS) for 4 h, followed by total RNA extraction using Trizol and complementary DNA synthesis via end-point RT-PCR (SuperScript III kit, Invitrogen) to investigate the expression of various genes including IL-8, NF-κB, MyD88, and TLR4. The oligonucleotides used are described in
Table 1 with actin as the housekeeping gene.
The evaluation of formazan formation through NBT reduction revealed the presence of formazan deposits in nHL-60 cells in the presence of PMA, whereas undifferentiated HL-60 cells did not exhibit such formations. Both peripheral blood neutrophils and HL-60 cells showed a 0.5-fold increase in IL-8 and IL-1β gene expression upon 100 ng/ml LPS stimulation (
Supplementary Fig. S1). These findings confirm that 1.3% DMSO induces functional changes in HL-60 cells, resulting in a neutrophil-like model (nHL-60) that serves as a valuable tool for investigating immune response mechanisms in human cells.
We then tested the impact of stimulation with 1 mM PA or 100 μM docosahexaenoic acid (DHA) for 4 h on the expression of IL-8, NF-κB, MyD88, and TLR4 was evaluated in nHL-60 cells, with 100 ng/ml LPS serving as a positive control of cell stimulation. Additionally, superoxide anion production was analyzed by incubating cells with 1 mg/ml NBT for 30 min, using 500 nM PMA as a positive control.
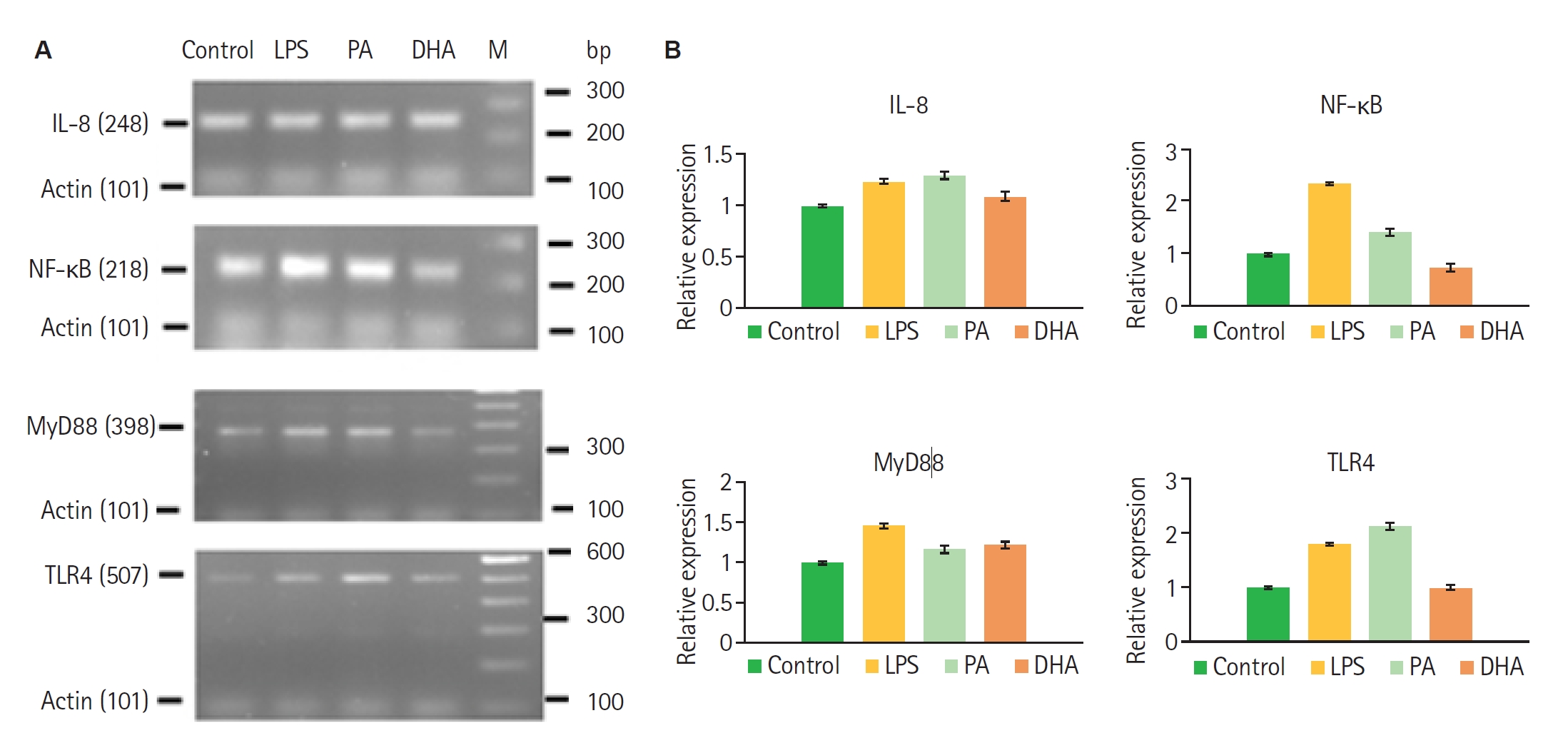
Gene expression in differentiated nHL-60 cells was assessed by band intensity following end-point RT-PCR. Pre-treatment with 1 mM PA resulted in increased relative expression levels of TLR4 (0.68), MyD88 (0.17), IL-8 (0.29), and NF-κB (0.42) compared to control, indicating upregulation of key components of the TLR4 signaling pathway. In contrast, treatment with 100 µM DHA, an unsaturated fatty acid, led to markedly reduced expression levels of TLR4 (0.02), MyD88 (0.22), IL-8 (0.09), and NF-κB, suggesting a suppressive effect on the pathway (
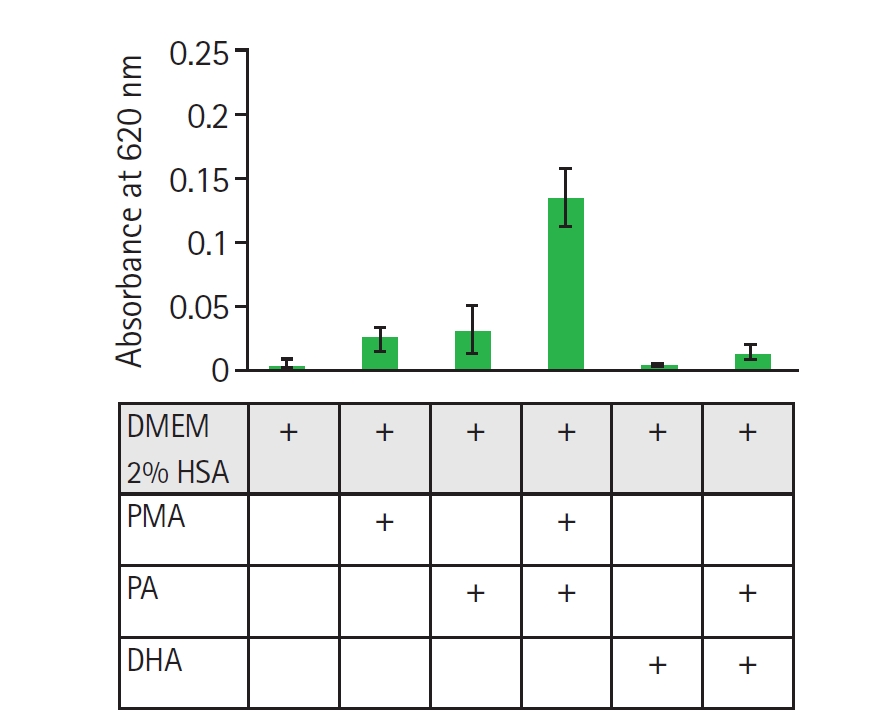
Fig. 1). Pre-treatment of nHL-60 cells for 30 min with fatty acids followed by PMA stimulation resulted in a significant increase in formazan absorbance after NBT reduction in the presence of PA, but not DHA. No such increase was observed in cells incubated solely with fatty acids, indicating that PA acts as an inducer of ROS in nHL-60 cells with a second stimulus (
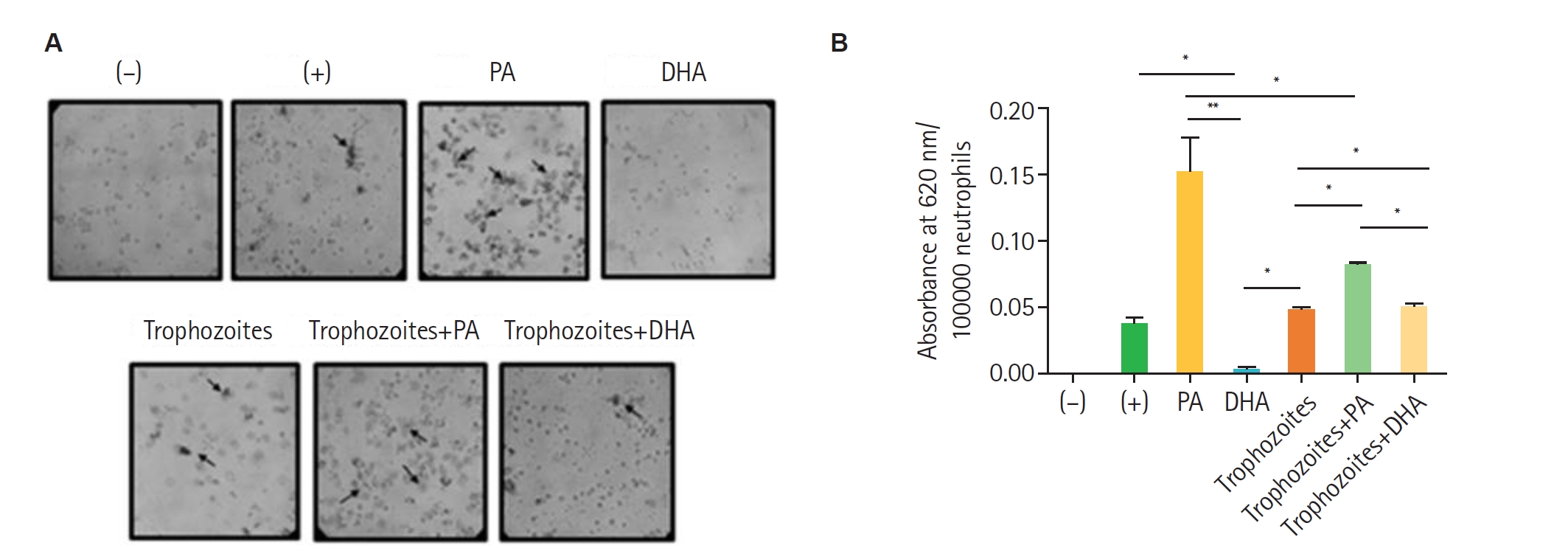
Fig. 2). The presence of 1 mM of PA enhanced the ROS production in human neutrophils (100,000 cells) in response to
T. vaginalis GT13 (10,000 trophozoites) compared with the 500 nM PMA positive control, while DHA had no effect. These findings suggest that PA can modulate the oxidative response of human neutrophils against
T. vaginalis, potentially affecting the efficiency of pathogen elimination (
Fig. 3). The activation of the NADPH oxidase complex depends on the phosphorylation of its components, which can be mediated by protein kinase C, promoting the translocation of enzyme subunits from the cytosol to the membrane. Both palmitic and stearic acids have been reported to activate NADPH oxidase in hepatocellular carcinoma cells [
14], and PA has been shown to do so through a protein kinase C–dependent pathway in aortic and endothelial cells [
3,
15]. These findings suggest the idea that PA induces ROS production in neutrophils.
Our results showing PA-induced ROS production are consistent with observations in other phagocytic cells, such as dendritic cells and macrophages [
16,
17]. Stearic acid—another abundant saturated fatty acid in plasma (~0.5 mM)—has also been reported to stimulate ROS production in human neutrophils at concentrations like those used for PA in this study [
10].
In the present work, PA triggered a stronger ROS response than
T. vaginalis, reinforcing the notion that saturated fatty acids can act as potent pro-oxidative stimuli. This agrees with previous evidence that palmitate activates TLR4–NOX2 signaling in macrophages, eliciting an oxidative burst comparable or superior to that induced by microbial ligands [
17].
Interestingly, although PA alone strongly induced ROS, co-incubation with
T. vaginalis suggested that the parasite may deploy protective mechanisms to limit oxidative damage (
Fig. 3B), as described for
Coxiella burnetii,
Helicobacter pylori, and
Francisella tularensis [
18]. These results suggest that
T. vaginalis could possess detoxification or ROS repair strategies to persist in the host environment.
To our knowledge, this is the first study to demonstrate the effects of PA on ROS production during
T. vaginalis–neutrophil interactions. Since both PA and the parasite induced ROS generation, they may converge on NADPH oxidase activation through TLR4 [
19], a receptor implicated in neutrophil oxidative responses. Nevertheless, alternative pathways could also be involved, as neutrophils may recognize PA and
T. vaginalis through distinct G protein-coupled receptors [
20].
Finally, high concentrations of neutrophil-recruiting mediators, including leukotrienes, monocyte chemoattractant proteins, and IL-8, have been detected in vaginal secretions. Since PA activates neutrophils, it would be relevant to evaluate whether PA stimulation enhances neutrophil migration and cytokine expression during T. vaginalis infection, better mimicking in vivo conditions.
In summary, our findings indicate that PA activates neutrophils and enhances ROS production in the context of T. vaginalis. These results have important implications for understanding how dietary fatty acids may modulate early innate responses against this parasite.
Notes
-
Author contributions
Conceptualization: Avila EE, Cuéllar-Mata P. Data curation: Sepúlveda-Angulo J. Formal analysis: Sepúlveda-Angulo J. Investigation: Sepúlveda-Angulo J. Methodology: Avila EE, Cuéllar-Mata P. Project administration: Avila EE, Cuéllar-Mata P. Resources: Olmos-Ortiz LM. Validation: Franco-Bárcenas B. Writing – original draft: Sepúlveda-Angulo J. Writing – review & editing: Franco-Bárcenas B, Avila EE, Cuéllar-Mata P.
-
Conflict of interest
The authors have no conflicts of interest to declare.
-
Acknowledgments
We thank Dr. Marion Brunck, UNAM, Mexico for their helpful discussions and suggestions. We also thank Dr Felipe Padilla-Vaca and Dr. Fernando Anaya-Velazquez for their kind donation of T. vaginalis GT21 strain. We are grateful to SICIHTI (before Consejo Nacional de Ciencia y Tecnología, Mexico) for a scholarship provided to JSA.
Supplementary information
Fig. 1.RT-PCR analysis of gene expression in nHL-60 cells stimulated with fatty acids. (A) nHL-60 cells were left unstimulated (Control) or treated with 100 ng/ml lipopolysaccharide (LPS), 1 mM palmitic acid (PA), or 100 µM docosahexaenoic acid (DHA) for 4 h. RNA was then extracted, and RT-PCR was performed. (B) Gene expression was normalized using actin as a housekeeping gene. Data represent mean±SD from 3 independent experiments (n=3). M, DNA size marker.
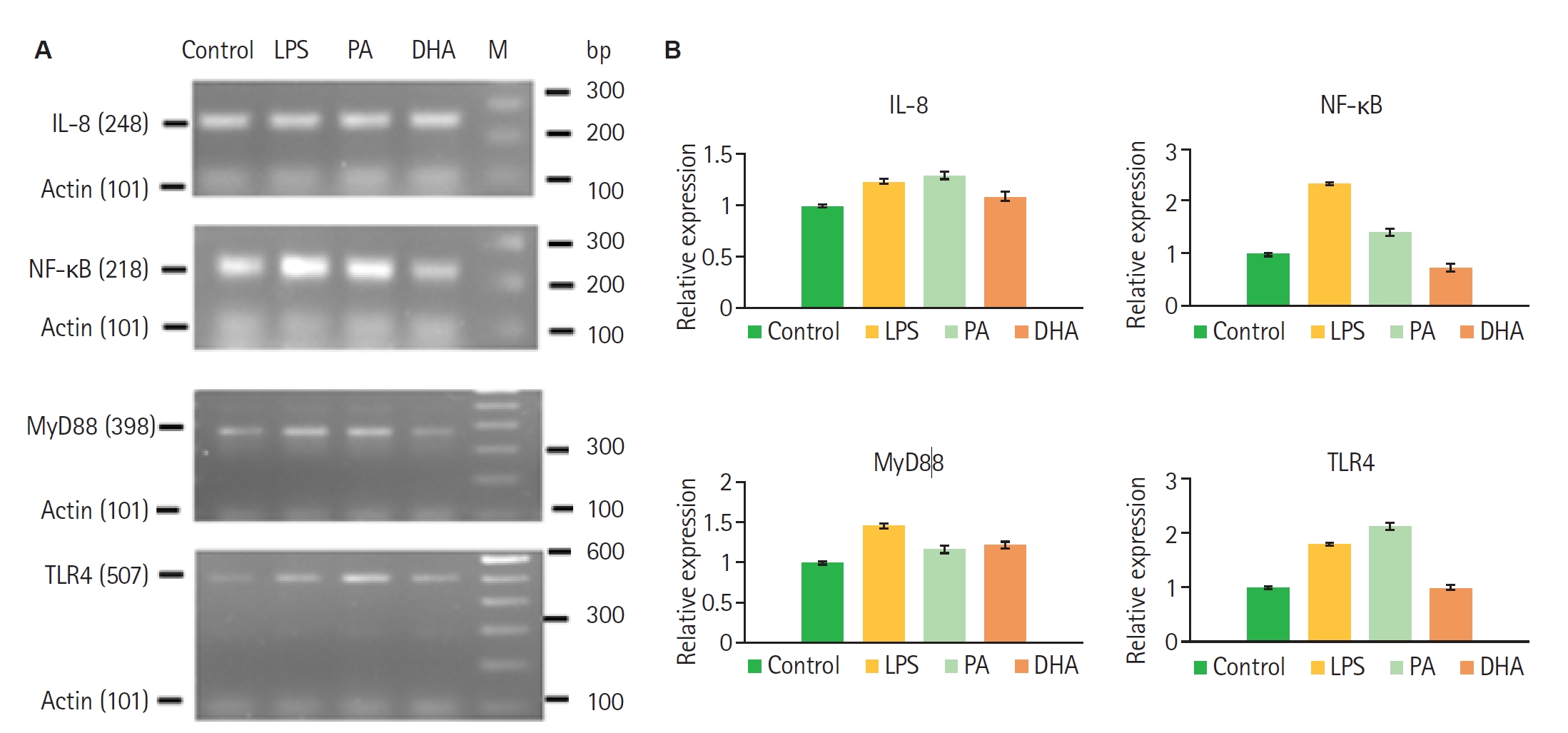
Fig. 2.Evaluation of reactive oxygen species generation via nitroblue tetrazolium reduction in nHL-60 cells treated with fatty acids. nHL-60 cells were incubated for 30 min with 1 mg/ml nitroblue tetrazolium in the presence or absence of 500 nM 12-myristate 13-acetate, 1 mM palmitic acid (PA), or 100 µM docosahexaenoic acid (DHA). Data represent mean±SD from 2 independent experiments (n=2), each performed in technical triplicate. DMEM, Dulbecco's modified eagle medium; HAS, human serum albumin.
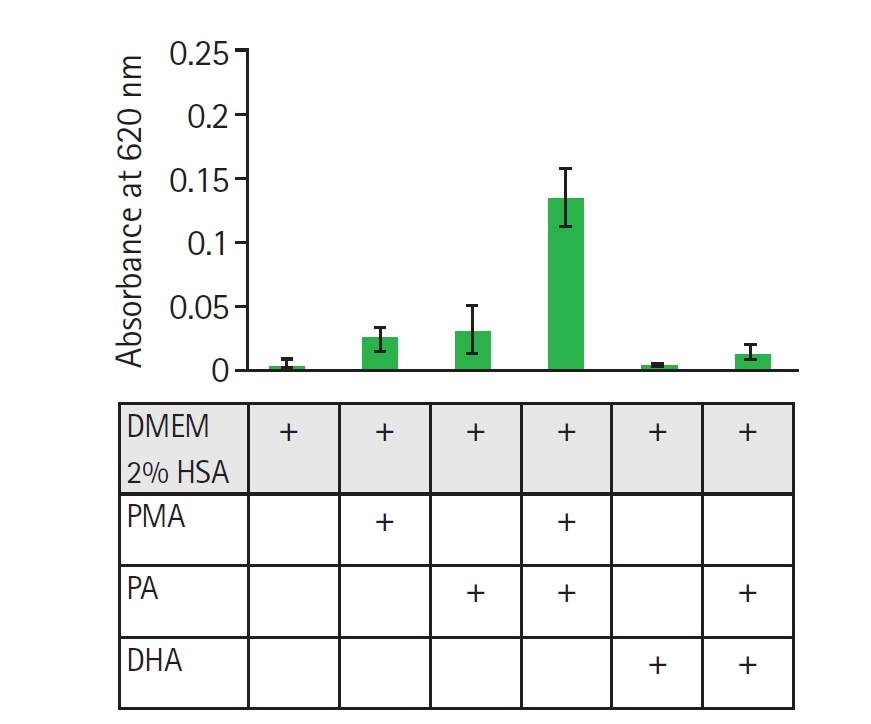
Fig. 3.Production of reactive oxygen species in neutrophils incubated with palmitic acid (PA) or docosahexaenoic acid (DHA) in the presence of Trichomonas vaginalis. Peripheral blood neutrophils (100,000) were isolated and incubated for 30 min with 1 mM PA or 100 µM DHA in the presence or absence of 10,000 trophozoites. Negative controls (unstimulated neutrophils) and positive controls (neutrophils stimulated with 500 nM 12-myristate 13-acetate) were included. Samples were analysed by (A) optical microscopy to detect formazan deposits and (B) quantification of reduced nitroblue tetrazolium as an indirect measure of formazan formation. Data represent mean±SD from 3 independent experiments (n=3), each performed in technical duplicate. Statistical analysis was performed using Kruskal-Wallis test with Dunn’s post hoc test. *P<0.05, **P<0.01.
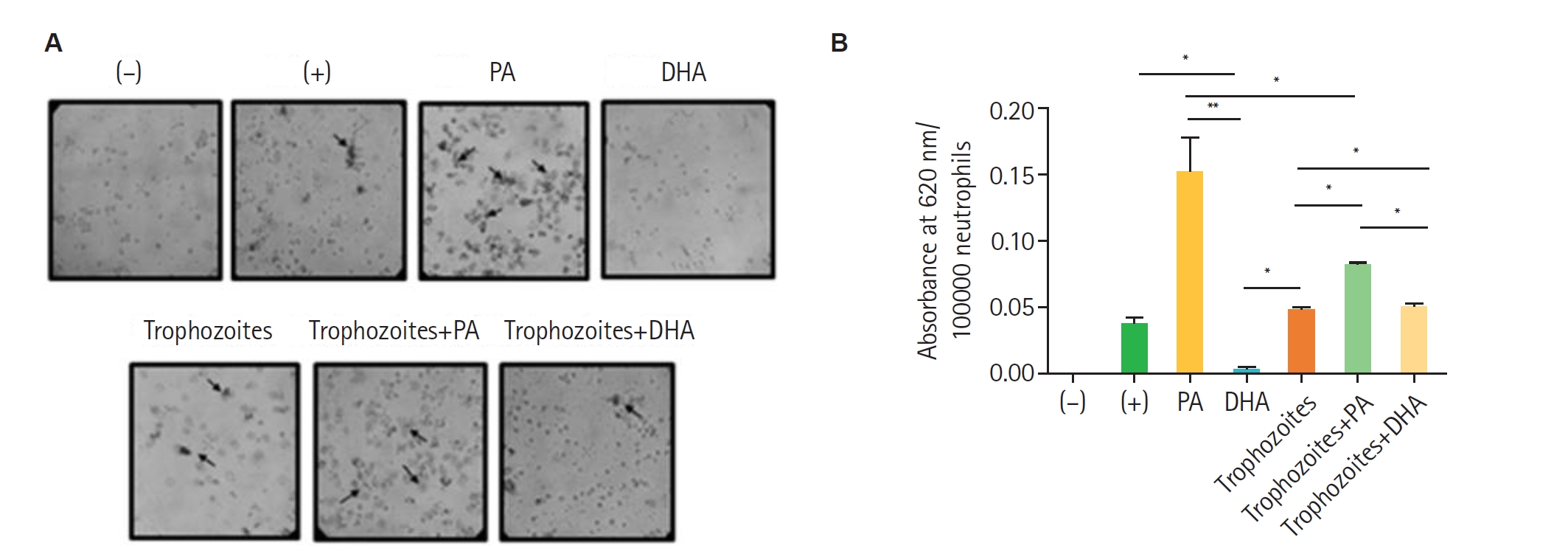
Table 1.Primer sequences used for RT-PCR amplification
Table 1.
|
Gene |
Forward primer |
Reverse primer |
Amplicon size of cDNA (bp) |
|
Actin |
TGAGATTGGCATGGCTTTATTTGT |
TGTCACCTTCACCGTCCAGTTT |
101 |
|
IL-1β |
CTGATGGCCCTAAACAGATGAAG |
GGTCGGAGATTCGTAGCAGCTGG |
87 |
|
TLR4 |
TGGATACGTTTCCTTATAAG |
GAAATGGAGGCACCCCTTC |
507 |
|
MyD88 |
GGCATATGCCTGAGCGTTCGA |
GGGGTTGGTGTAGTCGCAGACAGT |
398 |
|
NF-kB |
AAGTTCCTATAGAAGAGCAGCAGCGTGG |
TAGGAAGATCTCATCCCCACCGA |
218 |
|
IL-8 |
TTGGCAGCCTTCCTGATTTC |
AACTTCTCCACAACCCTCTGCA |
248 |
References
- 1. Akamatsu H, Niwa Y, Matsunaga K. Effect of palmitic acid on neutrophil functions in vitro. Int J Dermatol 2001;40:640-3. https://doi.org/10.1046/j.1365-4362.2001.01292.x
- 2. Nguyen MT, Favelyukis S, Nguyen AK, et al. A subpopulation of macrophages infiltrates hypertrophic adipose tissue and is activated by free fatty acids via Toll-like receptors 2 and 4 and JNK-dependent pathways. J Biol Chem 2007;282:35279-92. https://doi.org/10.1074/jbc.M706762200
- 3. Nicholas DA, Zhang K, Hung C, et al. Palmitic acid is a Toll-like receptor 4 ligand that induces human dendritic cell secretion of IL-1β. PLoS One 2017;12:e0176793. https://doi.org/10.1371/journal.pone.0176793
- 4. Korbecki J, Bajdak-Rusinek K. The effect of palmitic acid on inflammatory response in macrophages: an overview of molecular mechanisms. Inflamm Res 2019;68:915-32. https://doi.org/10.1007/s00011-019-01273-5
- 5. Snodgrass RG, Huang S, Namgaladze D, et al. Docosahexaenoic acid and palmitic acid reciprocally modulate monocyte activation in part through endoplasmic reticulum stress. J Nutr Biochem 2016;32:39-45. https://doi.org/10.1016/j.jnutbio.2016.01.010
- 6. Thomas CJ, Schroder K. Pattern recognition receptor function in neutrophils. Trends Immunol 2013;34:317-28. https://doi.org/10.1016/j.it.2013.02.008
- 7. Sabroe I, Dower SK, Whyte MK. The role of Toll-like receptors in the regulation of neutrophil migration, activation, and apoptosis. Clin Infect Dis 2005;41 Suppl 7:S421-6. https://doi.org/10.1086/431992
- 8. Jin J, Zhang X, Lu Z, et al. Acid sphingomyelinase plays a key role in palmitic acid-amplified inflammatory signaling triggered by lipopolysaccharide at low concentrations in macrophages. Am J Physiol Endocrinol Metab 2013;305:E856-67. https://doi.org/10.1152/ajpendo.00251.2013
- 9. Joshi-Barve S, Barve SS, Amancherla K, et al. Palmitic acid induces production of proinflammatory cytokine interleukin-8 from hepatocytes. Hepatology 2007;46:823-30. https://doi.org/10.1002/hep.21752
- 10. Wanten GJ, Janssen FP, Naber AH. Saturated triglycerides and fatty acids activate neutrophils depending on carbon chain-length. Eur J Clin Invest 2002;32:285-9. https://doi.org/10.1046/j.1365-2362.2002.00959.x
- 11. Fichorova RN. Impact of T. vaginalis infection on innate immune responses and reproductive outcome. J Reprod Immunol 2009;83:185-9. https://doi.org/10.1016/j.jri.2009.08.007
- 12. Mielczarek E, Blaszkowska J. Trichomonas vaginalis: pathogenicity and potential role in human reproductive failure. Infection 2016;44:447-58. https://doi.org/10.1007/s15010-015-0860-0
- 13. Song HO, Ryu JS. Superoxide anion production by human neutrophils activated by Trichomonas vaginalis. Korean J Parasitol 2013;51:479-84. https://doi.org/10.3347/kjp.2013.51.4.479
- 14. García-Ruiz I, Solís-Muñoz P, Fernández-Moreira D, et al. In vitro treatment of HepG2 cells with saturated fatty acids reproduces mitochondrial dysfunction found in nonalcoholic steatohepatitis. Dis Model Mech 2015;8:183-91. https://doi.org/10.1242/dmm.018234
- 15. Maloney E, Sweet IR, Hockenbery DM, et al. Activation of NF-kappaB by palmitate in endothelial cells: a key role for NADPH oxidase-derived superoxide in response to TLR4 activation. Arterioscler Thromb Vasc Biol 2009;29:1370-5. https://doi.org/10.1161/ATVBAHA.109.188813
- 16. Inoguchi T, Li P, Umeda F, et al. High glucose level and free fatty acid stimulate reactive oxygen species production through protein kinase C: dependent activation of NAD(P)H oxidase in cultured vascular cells. Diabetes 2000;49:1939-45. https://doi.org/10.2337/diabetes.49.11.1939
- 17. Kim SY, Jeong JM, Kim SJ, et al. Pro-inflammatory hepatic macrophages generate ROS through NADPH oxidase 2 via endocytosis of monomeric TLR4-MD2 complex. Nat Commun 2017;8:2247. https://doi.org/10.1038/s41467-017-02325-2
- 18. McCaffrey RL, Schwartz JT, Lindemann SR, et al. Multiple mechanisms of NADPH oxidase inhibition by type A and type B Francisella tularensis. J Leukoc Biol 2010;88:791-805. https://doi.org/10.1189/jlb.1209811
- 19. Zariffard MR, Harwani S, Novak RM, et al. Trichomonas vaginalis infection activates cells through toll-like receptor 4. Clin Immunol 2004;111:103-7. https://doi.org/10.1016/j.clim.2003.12.008
- 20. Katritch V, Cherezov V, Stevens RC. Structure-function of the G protein-coupled receptor superfamily. Annu Rev Pharmacol Toxicol 2013;53:531-56. https://doi.org/10.1146/annurev-pharmtox-032112-135923
Citations
Citations to this article as recorded by

 , Luis Mario Olmos-Ortiz2
, Luis Mario Olmos-Ortiz2 , Bernardo Franco-Bárcenas1
, Bernardo Franco-Bárcenas1 , Eva E. Avila1
, Eva E. Avila1 , Patricia Cuéllar-Mata1,*
, Patricia Cuéllar-Mata1,*