Abstract
The present study was performed to observe tegumental ultrastructure of Echinoparyphium recurvatum according to developmental stages. Worms (1, 3, 5 and 15-day old) were recovered from chicks experimentally infected with metacercariae from Radix auricularia coreana. One-day old worms were elongated and ventrally concave, and covered with peg-like tegumental spines except the adjacent areas of the head crown and excretory pore. Type I sensory papillae were distributed on the lip of the oral sucker, and grouped ciliated papillae were around the oral sucker. Peg-like tegumental spines were densely distributed on the anterior surface of the ventral sucker level. The ventral sucker had an aspinous tegument and no sensory papillae. Tegumental spines on the posterior surface of the ventral sucker level were sparsely distributed and disappeared posteriorly. In 3 and 5-day old worms, the tegument around the oral sucker was aspinose and wrinkled concentrically. The ventral sucker had a wrinkled tegument and many bulbous papillae. Type I sensory papillae were distributed between the bulbous papillae. Tegumental spines were spade-shaped with a terminal tip. A total of 45 collar spines including 4 end group ones on both ventral corners was alternately arranged in 2 rows. The 15-day old worms were very stout and their tegumental spines were tongue-shaped without a terminal tip. From the above results, it is confirmed that the surface ultrastructure of E. recurvatum was generally similar to that of other echinostomatid flukes. However, some features, i.e., morphological change of tegumental spines and appearence of sensory papillae on the ventral sucker according to development, and number, shape and arrangement of collar spines, were characteristic, which may be of taxonomic and bioecological significance.
-
Key words: Echinostomatidae, life cycle stages, scanning electron microscopy
INTRODUCTION
Echinoparyphium recurvatum (Trematoda: Echinostomatidae) is an intestinal digenean of wild birds and mammals including humans in Taiwan, Indonesia and Egypt (
Beaver et al., 1984). In Korea, adult worms are found in wild birds (
Chu et al., 1973) and a house rat (
Lee et al., 1990). Recently, its life history has been studied by Sohn (
1998) with biological and morphological characteristics.
Surface ultrastructure of trematodes gives a clue to the understanding of their morphological and taxonomical characteristics. Among the trematodes of the family Echinostomatidae, surface ultrastructural studies have been undertaken in various species (
Smales and Blankespoor, 1984;
Fried and Fujino, 1984;
Køie, 1987;
Lee et al., 1986,
1987,
1992;
Torii et al., 1989). However, study on the surface ultrastructure of
E. recurvatum have not yet been done. Therefore, in order to elucidate the ultrastructural characteristics of
E. recurvatum, worms recovered from chicks at 1, 3, 5 and 15 days after infection, were observed by scanning electron microscopy.
MATERIALS AND METHODS
The metacercariae of
E. recurvatum were isolated from
Radix auricularia coreana collected from Sunamchon, one of the streams in West Naktonggang (River), located in Kangso-gu, Pusan, Korea (
Sohn 1998). They were orally fed to 5 chicks using a gavage needle. Worms by developmental stages were recovered from chicks at 1, 3, 5 and 15 days after infection.
For scanning electron microscopy, worms were rinsed several times with physiological saline and 3 times with 0.1M cacodylate buffer (pH 7.2). They were fixed in 2.5% glutaraldehyde at 4℃, and post-fixed in 1% osmium tetroxide for 2hr at room temperature. After washing with 0.1M cacodylate buffer (pH 7.2), the specimens were dehydrated in a graded ethanol series (50%, 70%, 80%, 90%, 95% & absolute), dried in a ABT (model CP-5A) critical point dryer, and mounted on stubs. They were coated with gold using an ion sputtering coater (Bio-Rad, E 5300) and observed with a scanning electron microscope (ISI DS-130C, Korea) at an accelerating voltage of 10 kV.
RESULTS
One-day old worm
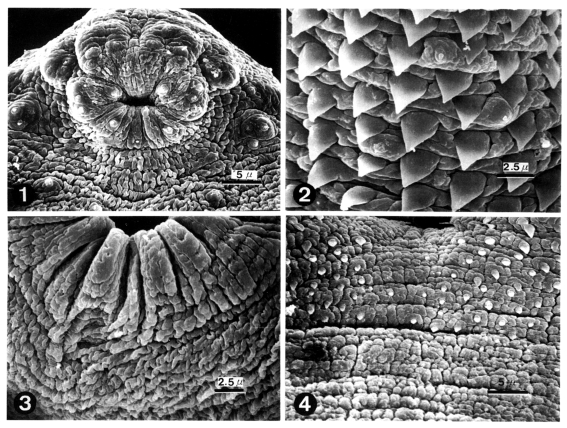
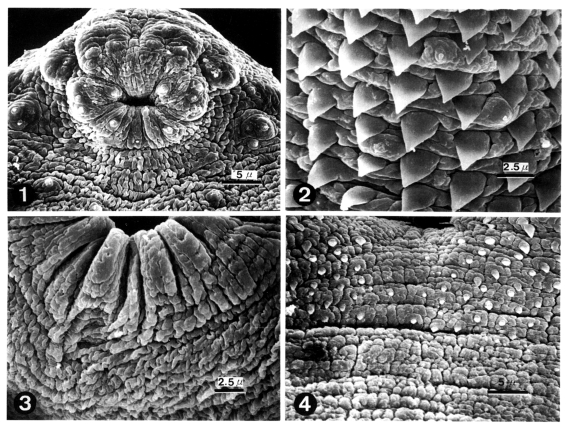
The body of worms was elongated and ventrally concave, and covered with peg-like tegumental spines except the adjacent areas of the head crown and excretory pore. The oral sucker had ciliated papillae (Type I sensory papillae) on the lip. Grouped ciliated papillae appeared around the oral sucker (
Fig. 1). The whole body surface was covered with cobblestone-like cytoplasmic processes, numerous tegumental spines and sensory papillae. Peg-like tegumental spines were densely distributed on the anterior surface of the ventral sucker level. Ciliated papillae appeared between the tegumental spines (
Fig. 2). The ventral sucker was located in the middle of the body, had aspinous tegument and lacked sensory papillae (
Fig. 3). Tegumental spines on the posterior surface of ventral sucker level were sparsely distributed and disappeared posteriorly (
Fig. 4).
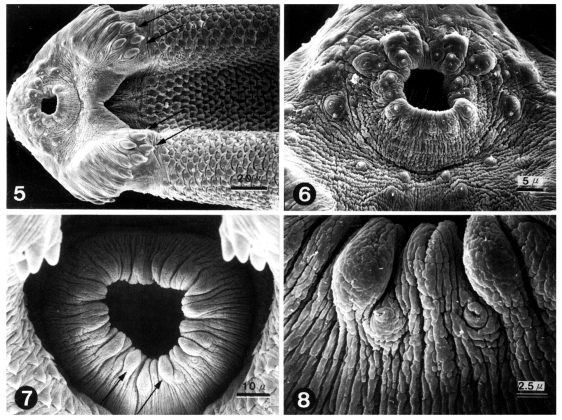
In the ventro-anterior region of worms, the oral sucker, head crown, 4 end group spines and peg-like tegumental spines were seen, and a ventral groove between the head crown and ventral sucker was formed (
Fig. 5). The tegument around the oral sucker was aspinose and wrinkled concentrically. Ciliated Type I sensory papillae were symmetrically distributed (
Fig. 6). The ventral sucker had a wrinkled tegument and many bulbous papillae. Type I sensory papillae were distributed between the bulbous papillae (
Figs. 7 & 8). On the dorso-anterior surface of the body, alternately arranged collar spines and regularly distributed tegumental spines were seen (
Fig. 9A). Tegumental spines were spade-shaped with a terminal tip. Type I sensory papillae were sparsely distributed between tegumental spines (
Fig. 9B).
The whole worm was concave and bent ventrally, and showed collar spines on the head crown and excretory pore in the posterior end (
Fig. 10). In the ventral surface anterior to the ventral sucker, numerous sensory papillae were scattered and tegumental spines were partly embedded in the cytoplasm (
Fig. 11). Collar spines were alternately arranged in 2 rows, those in the outer row were somewhat larger than in the inner row. The tegumental spines on the dorsal surface were regularly distributed at the level of the ventral sucker (
Fig. 12). On the dorsal surface anterior to the level of ventral sucker, spade-shaped tegumental spines with a terminal tip were regularly distributed and type I sensory papillae were sparsely scattered (
Figs. 13 & 14).
The whole worm was very stout and bore 45 collar spines including 4 end group ones on both ventral corners (
Fig. 15). The tegumental spines were tongue-shaped without a terminal tip. Type I sensory papillae were sparsely scattered between the tegumental spines (
Fig. 16).
DISCUSSION
Studies on the surface ultrastructure of echinostomatid flukes have been undertaken with various species i.e.
Isthmiophora melis (
Smale and Blankespoor, 1984),
Echinostoma revolutum (
Smale and Blankespoor, 1984;
Fried and Fujino, 1984),
Echinostoma hortense (
Lee et al., 1986;
Torii et al., 1989),
Mesorchis denticulatus (
Køie, 1987),
Echinochasmus japonicus (
Lee et al., 1987) and
Echinostoma cinetorchis (
Lee et al., 1992). However, the surface ultrastructure of trematodes belonging to the genus
Echinoparyphium was first observed in the present study.
The tegumental ultrastructure of
E. recurvatum was generally similar to that of other echinostomatid flukes in the shape of tegumental spines and cytoplasmic processes, and the distribution pattern of tegumental spines and sensory papillae (
Smale and Blankespoor, 1984;
Fried and Fujino, 1984;
Lee et al., 1986 &
1992;
Torii et al., 1989). However, some features such as the morphological change of tegumental spines and the appearence of sensory papillae on the ventral sucker according to development, and number, shape and arrangement of collar spines were characteristic, which may be of taxonomic and bioecological significance.
Similar to other echinostomatid digeneans,
E. recurvatum was covered with tegumental spines except the adjacent areas of the head crown and excretory pore. However, the shape of tegumental spines varied depending on the developmental stages. It was peg-like or triangular on the one-day old worms, on 3 and 5-day old juvenile worms was spade-shaped with a terminal tip, and on the 15-day old mature worms was tongue-shaped without a terminal tip. On the other hand, it has been known that tegumental spines on the juvenile worms of
E. revolutum and
E. cinetorchis are tongue or spade-shaped on the anterior to the ventral sucker and are peg-like on the posterior to the ventral sucker (
Fried and Fujino, 1984;
Lee et al., 1992). However, those on the adult echinostomatid flukes such as
I. melis,
E. revolutum,
E. hortense,
E. japonicus and
E. cinetorchis are tongue-shaped like those on the
E. recurvatum adult observed in this study (
Smale and Blankespoor, 1984;
Fried and Fujino, 1984;
Lee et al., 1986,
1987 &
1992).
The shape and distribution of tegumental spines of digeneans is closely related to worm maturation, parasitic niche, and whether they migrate or not. In case of some intestinal digeneans, tegumental spines are enlarged and more pointed as the worm maturates (
Køie, 1977;
Lee et al, 1985;
Hong et al, 1991;
Chai et al., 1998 &
2000). The aforementioned findings have been observed in some tissue migrating trematodes i.e.,
Fasciola hepatica (
Bennett, 1975),
Paragonimus westermani (
Lee et al., 1981) and
Paragonimus iloktsuenensis (
Lee et al., 1989). On the contrary, juveniles of
Clonorchis sinensis newly excysted in the duodenum have tegumental spines in the anterior half of body, but adults in the intrahepatic bile duct have not (
Fujino et al., 1979). In this study, tegmental spines of fully mature worms were larger and stouter than those of younger worms, but their distribution pattern did not change.
The number and arrangement of collar spines are very important in species identification of echinostomatid flukes. Among 7 species in the
Echinoparyphium group bearing 45 collar spines,
E. recurvatum is very similar to
E. mordwilkoi and
E. pseudorecurvatum. This fluke differs from two these species in the shape and size of the collar spines. The collar spines in
E. recurvatum are pointed, straight and shorter, whereas those in the other two species are bluntly ended and stumpy (
Kiseliene and Grabda-Kazubska, 1990;
Grabda-Kazubska and Kiseliene, 1991). The largest spine is the 4th one in
E. recurvetum, while it is the 2nd and 3rd one in
E. mordwilkoi and
E. pseudorecurvatum. These findings on the collar spines are in good agreement with those observed with SEM in the present study.
In the present study, two types of sensory papillae, uni-ciliated (type I) and roundly swollen (type II), were observed in
E. recurvatum. Type I papillae were mainly distributed on and around the oral sucker, in single or grouped forms, and sparsely between tegumental spines. Type II papillae were arranged on the ventral sucker of juvenile and adult worms. The function of these papillae is still unclear, although functions such as tango- and chemoreceptors have been speculated based on their shape and position on the body, and from the results of transmission electron microscopy showing sensory bulbs associated with both types of the papillae (
Morris and Threadgold, 1967;
Torii et al., 1989). As suggested for other digeneans, the papillae abundant on and around the oral sucker of
E. recurvatum are probably tactile sensory receptors involved in mechanisms for feeding and attachment.
References
- 1. Beaver PC, Jung RC, Cupp EW. Clinical Parasitology. 1984. 9th ed. Philadelphia, USA. Lea & Febiger; p 460.
- 2. Bennett CE. Scanning electron microscopy of Fasciola hepatica L. during growth and maturation in the mouse. J Parasitol 1975;61:892-898.
- 3. Chai JY, Guk SM, Han ET, et al. Surface ultrastructure of Metagonimus takahashii metacercariae and adults. Korean J Parasitol 2000;38:9-15.
- 4. Chai JY, Kang YJ, Choi SY, Guk SM, Yu JR, Lee SH. Surface ultrastructure of Metagonimus miyatai metacercariae and adults. Korean J Parasitol 1998;36:217-225.
- 5. Chu JK, Cho YJ, Chung SB, Won BO, Yoon MB. Study on the trematode parasites of the birds in Korea. Korean J Parasitol 1973;22:70-75. (in Korean).
- 6. Fried B, Fujino T. Scanning electron microscopy of Echinostoma revolutum (Trematoda) during development in the chick embryo and the domestic chick. Int J Parasitol 1984;14:75-81.
- 7. Fujino T, Ishii Y, Choi DW. Surface ultrastructure of the tegument of Clonorchis sinensis newly excysted juveniles and adult worms. J Parasitol 1979;65:579-590.
- 8. Grabda-Kazubska B, Kiseliene V. The life cycle of Echinoparyphium mordwilkoi Skrjabin, 1915 (Trematoda, Echinostomatidae). Acta Parasitol Polon 1991;36:167-173.
- 9. Hong SJ, Chai JY, Lee SH. Surface ultrastucture of the developmental stage of Heterophyopsis continua (Trematoda: Heterophyidae). J Parasitol 1991;77:613-620.
- 10. Kiseliene V, Grabda-Kazubska B. Echinoparyphium pseudorecurvatum sp. n. (Trematoda, Echinostomatidae) and its life cycle. Acta Parasisitol Polon 1990;35:285-295.
- 11. Køie M. Stereoscan studies of cercariae, metacercariae and adults of Cryptocotyle lingua (Creplin, 1825) Fischoeder, 1903 (Trematoda: Heterophyidae). J Parasitol 1977;63:835-839.
- 12. Køie M. Scanning electron microscopy of rediae, cercariae, metacercariae and adults of Mesorchis denticulatus (Rudolphi, 1802) (Trematoda: Echinostomatidae). Parasitol Res 1987;73:50-56.
- 13. Lee SH, Hong SJ, Chai JY, Hong ST, Seo BS. Tegumental ultrastructures of Echinostoma hortense observed by scanning electron microscopy. Korean J Parasitol 1986;24:63-70. (in Korean).
- 14. Lee SH, Hong SJ, Chai JY, Seo BS. Studies on intestinal trematodes in Korea. XV. Tegumental ultrastructures of Fibricola seoulensis according to developmental stages. Seoul J Med 1985;26:52-63.
- 15. Lee SH, Kim SJ, Chai JY, Sohn WM. Tegumental ultrastructure of Paragonimus iloktsuenensis according to the developmental stages. Korean J Parasitol 1989;27:57-66. (in Korean).
- 16. Lee SH, Jun HS, Sohn WM, Chai JY. Tegumental ultrastructure of juvenile and adult Echinostoma cinetorchis. Korean J Parasitol 1992;30:65-74. (in Korean).
- 17. Lee SH, Sohn WM, Chai JY. Echinostoma revolutum and Echinoparyphium recurvatum recovered from house rats in Yangyang-gun, Kangwon-do. Korean J Parasitol 1990;28:235-240.
- 18. Lee SH, Sohn WM, Hong ST. Scanning electron microscopical findings of Echinochasmus japonicus tegument. Korean J Parasitol 1987;25:51-58.
- 19. Lee SM, Hong ST, Lee SH, Shim TS. Ultrastructural study on the tegument of juvenile and adult Paragonimus westermani. Chung-Ang J Med 1981;6:579-585. (in Korean).
- 20. Morris GP, Threadgold LT. A presumed sensory structure associated with the tegument of Schistosoma mansoni. J Parasitol 1967;53:537-539.
- 21. Smales LR, Blankespoor HD. Echinostoma revolutum (Froelich, 1802) Looss, 1899 and Isthmiophora melis (Schrank, 1788) Luhe, 1909 (Echinostomatinae, Digenea): Scanning electron microscopy of the tegumental surfaces. J Helminthol 1984;58:187-195.
- 22. Sohn WM. Life history of Echinoparyphium recurvatum (Trematoda: Echinostomatidae) in Korea. Korean J Parasitol 1998;36:91-98.
- 23. Torii M, Tsuboi T, Hirai K, Nishida H. Ultrastructure of sensory receptors of adult Echinostoma hortense (Trematoda: Echinostomatidae). Jpn J Parasitol 1989;38:353-360.
Figs. 1-4
Scanning electron micrographs (SEM) of 1-day old worms of Echinoparyphium recurvatum.
Fig. 1. Ciliated papillae (Type I sensory papillae) seen in groups around the oral sucker and in single form on the lip. Fig. 2. Peg-like tegumental spines and type I sensory papillae on the ventro-lateral surface of the anterior half of the body. Fig. 3. Ventral sucker (VS) showing aspinous tegument and the absence of sensory papillae. Fig. 4. Peg-like spines distributed sparsely on the venral surface posterior to the ventral sucker disappeared posteriorly.
Figs. 5-8
SEM of 3-day old worms of Echinoparyphium recurvatum.
Fig. 5. Ventral view showing oral sucker, head crown and 4 end group spines (arrows). Fig. 6. Tegument around the oral sucker. Note the symmetrical distribution of sensory papillae and aspinose tegumental surface. Fig. 7. Ventral sucker showing wrinkled tegument and many bulbous papillae (arrows). Fig. 8. Enlargement of Fig. 7 showing type I sensory papillae between the bulbous papillae.

Figs. 9-12
SEM of 5-day old worms of Echinoparyphium recurvatum.
Fig. 9A. The dorsal anterior surface of the body showing alternately arranged collar spines and tegumental spines distributed to the level of the ventral sucker. Fig. 9B. Enlargement of a part of Fig. 9A showing spade-shaped tegumental spines and type I sensory papillae (arrow head).Fig. 10. A whole worm concave ventrally showing collar spines on the head crown and excretory pore (Ep) in the posterior end. Fig. 11. The ventral surface anterior to the ventral sucker showing numerous scattered sensory papillae (arrow heads) and tegumental spines partly embedded in the cytoplasm. Fig. 12. Collar spines and dorsal surface anterior to the level of the ventral sucker.

Figs. 13-16
SEM of 15-day old worms of Echinoparyphium recurvatum.
Fig. 13. Dorsal surface anterior to the level of the ventral sucker showing scattered type I sensory papillae (arrow heads) and spade-shaped tegumental spines. Fig. 14. Two type I sensory papillae (arrow heads) on the dorso-anterior surface. Fig. 15. A total of 45 collar spines including 4 end group spines on both ventral corners. Fig. 16. The dorsal surface anterior to the level of the ventral sucker showing tongue-shaped tegumental spines and type I sensory papillae (arrow heads).